- ▶
- Heaters/Source
- ▶
- Agilent Heaters and SensorsMass Spectrometry, Scientific Supplies & ManufacturingScientific Instrument Services 5973 Source Heater Tamper Resistant Allen Wrench 5973/5975 Quad Sensor 5985 Source Heater Assembly Agilent Interface Heater Assembly 5971 Interface Heater

- ▶
- Reference Material on InstrumentationArticle - A High Temperature Direct Probe for a Mass Spectrometer Design of a Direct Exposure Probe and Controller for use ona Hewlett-Packard 5989 Mass Spectrometer SIS AP1000 AutoProbe™ SIS AP2000 AutoProbe™ - Description of System HPP7: Direct Probe Electronics Console HPP7: Direct Probe for the Agilent (HP) 5973/5975 MSD HPP7: HP Direct Probe Application Notes HPP7: Installation Directions for the Direct Probe HPP7: Side Cover for the HP 5973 MSD HPP7: Support HPP7: Probe Inlet System for the Agilent (HP) 5973 and 5975 MSD with Automatic Indexed Stops HPP7: Theory of Operation of the Direct Probe and Probe Inlet System Direct Thermal Extraction Thermal Desorption Application Notes Environmental Thermal Desorption Application Notes Food Science Thermal Desorption Application Notes Forensic Thermal Desorption Application Notes GC Cryo-Trap Application Notes Headspace Application Notes Purge & Trap Thermal Desorption Application Notes Theory of Operation of the AutoDesorb® System AutoDesorb Notes for SIS Dealers Adsorbent Resin Application Notes Installation of the Short Path Thermal Desorption System on Agilent (HP) and Other GCs Installation of the Short Path Thermal Desorption System on a Varian 3400 GC AutoDesorb® System Development Team Thermal Desorption Applications and Reference Materials Installation of the Short Path Thermal Desorption System - TD5 Part I - Design & Operation of the Short Path ThermalDesorption System Installation Instructions for the Model 951 GC Cryo-Trap on the HP 5890 Series GC Installation Instructions for the Model 961 GC Cryo-Trap on the HP 5890 Series GC Operation of the Model 951/961 GC Cryo-Trap SIS GC Cryo Traps - Theory of Operation NIST/EPA/NIH Mass Spectral Enhancements - 1998 version (NIST98) SIMION 3D Ion Optics Class Mass Spectrometer Source Cleaning Methods MS Tip: Mass Spectrometer Source Cleaning Procedures Mass Spec Source Cleaning Procedures Micro-Mesh® Abrasive Sheets Research Papers Using New Era Syringe Pump Systems EI Positive Ion Spectra for Perfluorokerosene (PFK) Cap Liner Information How do I convert between fluid oz and milliliters? Which bottle material should I choose? Which bottle mouth should I choose? The Bottle Selection Guide CGA Connections for Gas Tanks Chemical Reaction Interface Mass Spectrometry (CRIMS)

- TD
- ▶
- AccessoriesTD Supply Kit Desorption Tubes Adsorbent Resins Desorption Tube Needles Desorption Tube Seals Desorption System Fittings GC Cryo-Trap Extraction Cell TD Sample Loader Prepacked, Conditioned Desorption Tubes Desorption Tube Packing Accessories Stainless Steel Purge Heads Injection Port Liners Tenax TA Poster TD Application Notes Customer Service

- LiteratureApplication Notes Adsorbent Resins Guide Mass Spec Tips SDS Sheets FAQ MS Calibration Compound Spectra Manuals MS Links/Labs/ Organizations MS Online Tools Flyers on Products/Services Scientific Supplies Catalog About Us NextAdvance Bullet Blender® Homogenizer Protocols Micro-Mesh® Literature Instrumentation Literature Agilent GC/MS Literature SIS News / E-Mail Newsletter NIST MS Database - Update Notifications

- ▶
- Thermal Desorption Applications and Reference MaterialsDirect Thermal Extraction Headspace Environmental Food Science Applications Pharmaceuticals Forensic Note 103: EPA Method 325B, Novel Thermal Desorption Instrument Modification to Improve Sensitivity Note 102: Identification of Contaminants in Powdered Beverages by Direct Extraction Thermal Desorption GC/MS Note 101: Identification of Contaminants in Powdered Foods by Direct Extraction Thermal Desorption GC/MS Note 100: Volatile and Semi-Volatile Profile Comparison of Whole Versus Cracked Versus Dry Homogenized Barley Grains by Direct Thermal Extraction Note 99: Volatile and Semi-Volatile Profile Comparison of Whole vs. Dry Homogenized Wheat, Rye and Barley Grains by Direct Thermal Extraction GC/MS Note 98: Flavor and Aroma Profiles of Truffle Oils by Thermal Desorption GC/MS Note 97: Flavor Profiles of Imported and Domestic Beers by Purge & Trap Thermal Desorption GC/MS Note 95: Detection of Explosives on Clothing Material by Direct and AirSampling Thermal Desorption GC/MS Note 94: Detection of Nepetalactone in the Nepeta Cataria Plant by Thermal Desorption GC/MS Note 93: Detection of Benzene in Carbonated Beverages with Purge & Trap Thermal Desorption GC/MS Note 88: Analysis of Silicone Contaminants on Electronic Components by Thermal Desorption GC-MS Note 84: Vacuum Pump Exhaust Filters - Charcoal Exhaust Traps Note 83: Vacuum Pump Exhaust Filters - Oil Mist Eliminators Note 82: Vacuum Pump Exhaust Filters Note 80: Design, Development and Testing of a Microprocessor ControlledAutomated Short Path Thermal Desorption Apparatus Note 79: Volatile Organic Compounds From Electron Beam Cured and Partially Electron Beam Cured Packaging Using Automated Short Path Thermal Desorption Note 77: The Determination of Volatile Organic Compounds in VacuumSystem Components Note 75: An Apparatus for Sampling Volatile Organics From LivePlant Material Using Short Path Thermal Desorption Note 73: The Analysis of Perfumes and their Effect on Indoor Air Pollution Note 71: Flavor Profile Determination of Rice Samples Using Shor tPath Thermal Desorption GC Methods Note 65: Determination of Ethylene by Adsorbent Trapping and Thermal Desorption - Gas Chromatography Note 64: Comparison of Various GC/MS Techniques For the Analysis of Black Pepper (Piper Nigrum) Note 63: Determination of Volatile and Semi-Volatile Organics in Printer Toners Using Thermal Desorption GC Techniques Note 60: Programmable Temperature Ramping of Samples Analyzed ViaDirect Thermal Extraction GC/MS Note 57: Aroma Profiles of Lavandula species Note 55: Seasonal Variation in Flower Volatiles Note 54: Identification of Volatile Organic Compounds in Office Products Note 43: Volatile Organic Composition In Blueberries Note 42: The Influence of Pump Oil Purity on Roughing Pumps Note 41: Hydrocarbon Production in Pine by Direct Thermal Extraction Note 40: Comparison of Septa by Direct Thermal Extraction Note 39: Comparison of Sensitivity Of Headspace GC, Purge and Trap Thermal Desorption and Direct Thermal Extraction Techniques For Volatile Organics Note 38: A New Micro Cryo-Trap For Trapping Of Volatiles At the Front Of a GC Capillary Column Note 37: Volatile Organic Emissions from Automobile Tires Note 36: Identification Of Volatile Organic Compounds In a New Automobile Note 35: Volatile Organics Composition of Cranberries Note 34: Selection Of Thermal Desorption and Cryo-Trap Parameters In the Analysis Of Teas Note 33: Changes in Volatile Organic Composition in Milk Over Time Note 32: Selection and Use of Adsorbent Resins for Purge and Trap Thermal Desorption Applications Note 31: Volatile Organic Composition in Several Cultivars of Peaches Note 30: Comparison Of Cooking Oils By Direct Thermal Extraction and Purge and Trap GC/MS Note 29: Analysis Of Volatile Organics In Oil Base Paints By Automated Headspace Sampling and GC Cryo-Focusing Note 28: Analysis Of Volatile Organics In Latex Paints By Automated Headspace Sampling and GC Cryo-Focusing Note 27: Analysis of Volatile Organics In Soils By Automated Headspace GC Note 26: Volatile Organics Present in Recycled Air Aboard a Commercial Airliner Note 25: Flavor and Aroma in Natural Bee Honey Note 24: Selection of GC Guard Columns For Use With the GC Cryo-Trap Note 23: Frangrance Qualities in Colognes Note 22: Comparison Of Volatile Compounds In Latex Paints Note 21: Detection and Identification Of Volatile and Semi-Volatile Organics In Synthetic Polymers Used In Food and Pharmaceutical Packaging Note 20: Using Direct Thermal Desorption to Assess the Potential Pool of Styrene and 4-Phenylcyclohexene In Latex-Backed Carpets Note 19: A New Programmable Cryo-Cooling/Heating Trap for the Cryo-Focusing of Volatiles and Semi-Volatiles at the Head of GC Capillary Columns Note 18: Determination of Volatile Organic Compounds In Mushrooms Note 17: Identification of Volatile Organics in Wines Over Time Note 16: Analysis of Indoor Air and Sources of Indoor Air Contamination by Thermal Desorption Note 14: Identification of Volatiles and Semi-Volatiles In Carbonated Colas Note 13: Identification and Quantification of Semi-Volatiles In Soil Using Direct Thermal Desorption Note 12: Identification of the Volatile and Semi-Volatile Organics In Chewing Gums By Direct Thermal Desorption Note 11: Flavor/Fragrance Profiles of Instant and Ground Coffees By Short Path Thermal Desorption Note 10: Quantification of Naphthalene In a Contaminated Pharmaceutical Product By Short Path Thermal Desorption Note 9: Methodologies For the Quantification Of Purge and Trap Thermal Desorption and Direct Thermal Desorption Analyses Note 8: Detection of Volatile Organic Compounds In Liquids Utilizing the Short Path Thermal Desorption System Note 7: Chemical Residue Analysis of Pharmaceuticals Using The Short Path Thermal Desorption System Note 6: Direct Thermal Analysis of Plastic Food Wraps Using the Short Path Thermal Desorption System Note 5: Direct Thermal Analysis Using the Short Path Thermal Desorption System Note 4: Direct Analysis of Spices and Coffee Note 3: Indoor Air Pollution Note 2: Detection of Arson Accelerants Using Dynamic Headspace with Tenax® Cartridges Thermal Desorption and Cryofocusing Note 1: Determination of Off-Odors and Other Volatile Organics In Food Packaging Films By Direct Thermal Analysis-GC-MS

- ▶
- Environmental Thermal Desorption Application NotesNote 42: The Influence of Pump Oil Purity on Roughing Pumps Note 41: Hydrocarbon Production in Pine by Direct Thermal Extraction Note 40: Comparison of Septa by Direct Thermal Extraction Note 39: Comparison of Sensitivity Of Headspace GC, Purge and Trap Thermal Desorption and Direct Thermal Extraction Techniques For Volatile Organics Note 38: A New Micro Cryo-Trap For Trapping Of Volatiles At the Front Of a GC Capillary Column Note 37: Volatile Organic Emissions from Automobile Tires Note 36: Identification Of Volatile Organic Compounds In a New Automobile Note 27: Analysis of Volatile Organics In Soils By Automated Headspace GC Note 26: Volatile Organics Present in Recycled Air Aboard a Commercial Airliner Note 16: Analysis of Indoor Air and Sources of Indoor Air Contamination by Thermal Desorption Note 13: Identification and Quantification of Semi-Volatiles In Soil Using Direct Thermal Desorption Note 8: Detection of Volatile Organic Compounds In Liquids Utilizing the Short Path Thermal Desorption System Note 3: Indoor Air Pollution Note 2: Detection of Arson Accelerants Using Dynamic Headspace with Tenax® Cartridges Thermal Desorption and Cryofocusing

- Direct Thermal Extraction Thermal Desorption Application NotesNote 103: EPA Method 325B, Novel Thermal Desorption Instrument Modification to Improve Sensitivity Note 102: Identification of Contaminants in Powdered Beverages by Direct Extraction Thermal Desorption GC/MS Note 101: Identification of Contaminants in Powdered Foods by Direct Extraction Thermal Desorption GC/MS Note 100: Volatile and Semi-Volatile Profile Comparison of Whole Versus Cracked Versus Dry Homogenized Barley Grains by Direct Thermal Extraction Note 99: Volatile and Semi-Volatile Profile Comparison of Whole vs. Dry Homogenized Wheat, Rye and Barley Grains by Direct Thermal Extraction GC/MS Note 98: Flavor and Aroma Profiles of Truffle Oils by Thermal Desorption GC/MS Note 95: Detection of Explosives on Clothing Material by Direct and AirSampling Thermal Desorption GC/MS Note 94: Detection of Nepetalactone in the Nepeta Cataria Plant by Thermal Desorption GC/MS Note 41: Hydrocarbon Production in Pine by Direct Thermal Extraction Note 40: Comparison of Septa by Direct Thermal Extraction Note 39: Comparison of Sensitivity Of Headspace GC, Purge and Trap Thermal Desorption and Direct Thermal Extraction Techniques For Volatile Organics Note 37: Volatile Organic Emissions from Automobile Tires Note 30: Comparison Of Cooking Oils By Direct Thermal Extraction and Purge and Trap GC/MS Note 21: Detection and Identification Of Volatile and Semi-Volatile Organics In Synthetic Polymers Used In Food and Pharmaceutical Packaging Note 20: Using Direct Thermal Desorption to Assess the Potential Pool of Styrene and 4-Phenylcyclohexene In Latex-Backed Carpets Note 13: Identification and Quantification of Semi-Volatiles In Soil Using Direct Thermal Desorption Note 12: Identification of the Volatile and Semi-Volatile Organics In Chewing Gums By Direct Thermal Desorption Note 11: Flavor/Fragrance Profiles of Instant and Ground Coffees By Short Path Thermal Desorption Note 10: Quantification of Naphthalene In a Contaminated Pharmaceutical Product By Short Path Thermal Desorption Note 9: Methodologies For the Quantification Of Purge and Trap Thermal Desorption and Direct Thermal Desorption Analyses Note 7: Chemical Residue Analysis of Pharmaceuticals Using The Short Path Thermal Desorption System Note 6: Direct Thermal Analysis of Plastic Food Wraps Using the Short Path Thermal Desorption System Note 5: Direct Thermal Analysis Using the Short Path Thermal Desorption System Note 4: Direct Analysis of Spices and Coffee Note 1: Determination of Off-Odors and Other Volatile Organics In Food Packaging Films By Direct Thermal Analysis-GC-MS

- Application NotesNote 103: EPA Method 325B, Novel Thermal Desorption Instrument Modification to Improve Sensitivity Note 102: Identification of Contaminants in Powdered Beverages by Direct Extraction Thermal Desorption GC/MS Note 101: Identification of Contaminants in Powdered Foods by Direct Extraction Thermal Desorption GC/MS Note 100: Volatile and Semi-Volatile Profile Comparison of Whole Versus Cracked Versus Dry Homogenized Barley Grains by Direct Thermal Extraction Note 99: Volatile and Semi-Volatile Profile Comparison of Whole vs. Dry Homogenized Wheat, Rye and Barley Grains by Direct Thermal Extraction GC/MS Note 98: Flavor and Aroma Profiles of Truffle Oils by Thermal Desorption GC/MS Note 97: Flavor Profiles of Imported and Domestic Beers by Purge & Trap Thermal Desorption GC/MS Note 96: Reducing Warping in Mass Spectrometer Filaments, with SISAlloy® Yttria/Rhenium Filaments Note 95: Detection of Explosives on Clothing Material by Direct and AirSampling Thermal Desorption GC/MS Note 94: Detection of Nepetalactone in the Nepeta Cataria Plant by Thermal Desorption GC/MS Note 93: Detection of Benzene in Carbonated Beverages with Purge & Trap Thermal Desorption GC/MS Note 92: Yttria Coated Mass Spectrometer Filaments Note 91: AutoProbe DEP Probe Tip Temperatures Note 90: An Automated MS Direct Probe for use in an Open Access Environment Note 89: Quantitation of Organics via a Mass Spectrometer Automated Direct Probe Note 88: Analysis of Silicone Contaminants on Electronic Components by Thermal Desorption GC-MS Note 87: Design and Development of an Automated Direct Probe for a Mass Spectrometer Note 86: Simulation of a Unique Cylindrical Quadrupole Mass Analyzer Using SIMION 7.0. Note 85: Replacing an Electron Multiplier in the Agilent (HP) 5973 MSD Note 84: Vacuum Pump Exhaust Filters - Charcoal Exhaust Traps Note 83: Vacuum Pump Exhaust Filters - Oil Mist Eliminators Note 82: Vacuum Pump Exhaust Filters Note 81: Rapid Bacterial Chemotaxonomy By DirectProbe/MSD Note 80: Design, Development and Testing of a Microprocessor ControlledAutomated Short Path Thermal Desorption Apparatus Note 79: Volatile Organic Compounds From Electron Beam Cured and Partially Electron Beam Cured Packaging Using Automated Short Path Thermal Desorption Note 78: A New Solution to Eliminate MS Down-Time With No-Tool-Changing of Analytical GC Columns Note 77: The Determination of Volatile Organic Compounds in VacuumSystem Components Note 76: Determination of the Sensitivity of a CRIMS System Note 75: An Apparatus for Sampling Volatile Organics From LivePlant Material Using Short Path Thermal Desorption Note 74: Examination of Source Design in Electrospray-TOF Using SIMION 3D Note 73: The Analysis of Perfumes and their Effect on Indoor Air Pollution Note 72: 1998 Version of the NIST/EPA/NIH Mass Spectral Library, NIST98 Note 71: Flavor Profile Determination of Rice Samples Using Shor tPath Thermal Desorption GC Methods Note 70: Application of SIMION 6.0 To a Study of the Finkelstein Ion Source: Part II Note 69: Application of SIMION 6.0 To a Study of the Finkelstein Ion Source: Part 1 Note 68: Use of a PC Plug-In UV-Vis Spectrometer To Monitor the Plasma Conditions In GC-CRIMS Note 67: Using Chemical Reaction Interface Mass Spectrometry (CRIMS) To Monitor Bacterial Transport In In Situ Bioremediation Note 66: Probe Tip Design For the Optimization of Direct Insertion Probe Performance Note 65: Determination of Ethylene by Adsorbent Trapping and Thermal Desorption - Gas Chromatography Note 64: Comparison of Various GC/MS Techniques For the Analysis of Black Pepper (Piper Nigrum) Note 63: Determination of Volatile and Semi-Volatile Organics in Printer Toners Using Thermal Desorption GC Techniques Note 62: Analysis of Polymer Samples Using a Direct Insertion Probe and EI Ionization Note 61: Analysis of Sugars Via a New DEP Probe Tip For Use With theDirect Probe On the HP5973 MSD Note 60: Programmable Temperature Ramping of Samples Analyzed ViaDirect Thermal Extraction GC/MS Note 59: Computer Modeling of a TOF Reflectron With Gridless Reflector Using SIMION 3D Note 58: Direct Probe Analysis and Identification of Multicomponent Pharmaceutical Samples via Electron Impact MS Note 57: Aroma Profiles of Lavandula species Note 56: Mass Spec Maintenance & Cleaning Utilizing Micro-Mesh® Abrasive Sheets Note 55: Seasonal Variation in Flower Volatiles Note 54: Identification of Volatile Organic Compounds in Office Products Note 53: SIMION 3D v6.0 Ion Optics Simulation Software Note 52: Computer Modeling of Ion Optics in Time-of-Flight mass Spectrometry Using SIMION 3D Note 51: Development and Characterization of a New Chemical Reaction Interface for the Detection of Nonradioisotopically Labeled Analytes Using Mass Spectrometry (CRIMS) Note 50: The Analysis of Multiple Component Drug Samples Using a Direct Probe Interfaced to the HP 5973 MSD Note 49: Analysis of Cocaine Utilizing a New Direct Insertion Probe on a Hewlett Packard 5973 MSD Note 48: Demonstration of Sensitivity Levels For the Detection of Caffeine Using a New Direct Probe and Inlet for the HP 5973 MSD Note 47: The Application Of SIMION 6.0 To Problems In Time-of-Flight Mass Spectrometry Note 46: Delayed Extraction and Laser Desorption: Time-lag Focusing and Beyond Note 45: Application of SIMION 6.0 to Filament Design for Mass Spectrometer Ionization Sources Note 44: The Design Of a New Direct Probe Inlet For a Mass Spectrometer Note 43: Volatile Organic Composition In Blueberries Note 42: The Influence of Pump Oil Purity on Roughing Pumps Note 41: Hydrocarbon Production in Pine by Direct Thermal Extraction Note 40: Comparison of Septa by Direct Thermal Extraction Note 39: Comparison of Sensitivity Of Headspace GC, Purge and Trap Thermal Desorption and Direct Thermal Extraction Techniques For Volatile Organics Note 38: A New Micro Cryo-Trap For Trapping Of Volatiles At the Front Of a GC Capillary Column Note 37: Volatile Organic Emissions from Automobile Tires Note 36: Identification Of Volatile Organic Compounds In a New Automobile Note 35: Volatile Organics Composition of Cranberries Note 34: Selection Of Thermal Desorption and Cryo-Trap Parameters In the Analysis Of Teas Note 33: Changes in Volatile Organic Composition in Milk Over Time Note 32: Selection and Use of Adsorbent Resins for Purge and Trap Thermal Desorption Applications Note 31: Volatile Organic Composition in Several Cultivars of Peaches Note 30: Comparison Of Cooking Oils By Direct Thermal Extraction and Purge and Trap GC/MS Note 29: Analysis Of Volatile Organics In Oil Base Paints By Automated Headspace Sampling and GC Cryo-Focusing Note 28: Analysis Of Volatile Organics In Latex Paints By Automated Headspace Sampling and GC Cryo-Focusing Note 27: Analysis of Volatile Organics In Soils By Automated Headspace GC Note 26: Volatile Organics Present in Recycled Air Aboard a Commercial Airliner Note 25: Flavor and Aroma in Natural Bee Honey Note 24: Selection of GC Guard Columns For Use With the GC Cryo-Trap Note 23: Frangrance Qualities in Colognes Note 22: Comparison Of Volatile Compounds In Latex Paints Note 21: Detection and Identification Of Volatile and Semi-Volatile Organics In Synthetic Polymers Used In Food and Pharmaceutical Packaging Note 20: Using Direct Thermal Desorption to Assess the Potential Pool of Styrene and 4-Phenylcyclohexene In Latex-Backed Carpets Note 19: A New Programmable Cryo-Cooling/Heating Trap for the Cryo-Focusing of Volatiles and Semi-Volatiles at the Head of GC Capillary Columns Note 18: Determination of Volatile Organic Compounds In Mushrooms Note 17: Identification of Volatile Organics in Wines Over Time Note 16: Analysis of Indoor Air and Sources of Indoor Air Contamination by Thermal Desorption Note 14: Identification of Volatiles and Semi-Volatiles In Carbonated Colas Note 13: Identification and Quantification of Semi-Volatiles In Soil Using Direct Thermal Desorption Note 12: Identification of the Volatile and Semi-Volatile Organics In Chewing Gums By Direct Thermal Desorption Note 11: Flavor/Fragrance Profiles of Instant and Ground Coffees By Short Path Thermal Desorption Note 10: Quantification of Naphthalene In a Contaminated Pharmaceutical Product By Short Path Thermal Desorption Note 9: Methodologies For the Quantification Of Purge and Trap Thermal Desorption and Direct Thermal Desorption Analyses Note 8: Detection of Volatile Organic Compounds In Liquids Utilizing the Short Path Thermal Desorption System Note 7: Chemical Residue Analysis of Pharmaceuticals Using The Short Path Thermal Desorption System Note 6: Direct Thermal Analysis of Plastic Food Wraps Using the Short Path Thermal Desorption System Note 5: Direct Thermal Analysis Using the Short Path Thermal Desorption System Note 4: Direct Analysis of Spices and Coffee Note 3: Indoor Air Pollution Note 2: Detection of Arson Accelerants Using Dynamic Headspace with Tenax® Cartridges Thermal Desorption and Cryofocusing Note 1: Determination of Off-Odors and Other Volatile Organics In Food Packaging Films By Direct Thermal Analysis-GC-MS Tech No. "A" Note 14: Elimination of "Memory" Peaks in Thermal Desorption Improving Sensitivity in the H.P. 5971 MSD and Other Mass Spectrometers - Part I of II Improving Sensitivity in the H.P. 5971 MSD and Other Mass Spectrometers- Part II of II Adsorbent Resins Guide Development and Field Tests of an Automated Pyrolysis Insert for Gas Chromatography. Hydrocarbon Production in Pine by Direct Thermal Extraction A New Micro Cryo-Trap for the Trapping of Volatiles at the Front of a GC Capillary (019P) - Comparison of Septa by Direct Thermal Extraction Volatile Organic Composition in Blueberry Identification of Volatile Organic Compounds in Office Products Detection and Indentification of Volatiles in Oil Base Paintsby Headspace GC with On Column Cryo-Trapping Evaluation of Septa Using a Direct Thermal Extraction Technique INFLUENCE OF STORAGE ON BLUEBERRY VOLATILES Selection of Thermal Desorption and Cryo-Trap Parameters in the Analysis of Teas Redesign and Performance of a Diffusion Based Solvent Removal Interface for LC/MS The Design of a New Direct Probe Inlet for a Mass Spectrometer Analytes Using Mass Spectrometry (CRIMS) Application of SIMION 6.0 to Filament Design for Mass Spectrometer Ionization Sources A Student Guide for SIMION Modeling Software Application of SIMION 6.0 to Problems in Time-of-flight Mass Spectrometry Comparison of Sensitivity of Headspace GC, Purge and TrapThermal Desorption and Direct Thermal Extraction Techniques forVolatile Organics The Influence of Pump Oil Purity on Roughing Pumps Analysis of Motor Oils Using Thermal Desorption-Gas Chromatography-Mass Spectrometry IDENTIFICATION OF VOLATILE ORGANIC COMPOUNDS IN PAPER PRODUCTS Computer Modeling of Ion Optics in Time-of-Flight mass Spectrometry using SIMION 3D Seasonal Variation in Flower Volatiles Development of and Automated Microprocessor Controlled Gas chromatograph Fraction Collector / Olfactometer Delayed Extraction and Laser Desorption: Time-lag Focusing and Beyond A New Micro Cryo-Trap for the Trapping of Volatiles at the Front of a GC Column Design of a Microprocessor Controlled Short Path Thermal Desorption Autosampler Computer Modeling of Ion Optics in Time-of-Flight Mass Spectrometry Using SIMION 3D Thermal Desorption Instrumentation for Characterization of Odors and Flavors

- ▶
- Note 13: Identification and Quantification of Semi-Volatiles In Soil Using Direct Thermal Desorption (This Page)
by John J. Manura
INTRODUCTION
New methods are needed for the analyses of semi-volatile organic compounds from complex matrices such as: soils and sludge from hazardous dump sites and environmental spills. Conventional methods incorporate solvent extraction which is both time consuming and requires the use of large quantities of organic solvents. Not only is there concern over the safe handling of these toxic and carcinogenic solvents, but their safe disposal is becoming increasingly expensive and troublesome. In addition, during the extraction process, the solvent extractions must be concentrated. In this concentration process, the solvent is usually evaporated and exhausted in a laboratory fume hood, thereby contributing to an additional environmental problem. Other techniques, as Super Critical Fluid Extraction (SFE) and Thermal Desorption, have recently become acceptable for the extraction of semi-volatiles from soil and other solid matrices (1,2 & 3). SFE still requires a small amount of solvent and about 1 hour for sample preparation. In this article, a new technique encompassing the Short Path Thermal Desorption System interfaced to a GC/MS and using the Direct Thermal Desorption Technique is used to identify and quantify the semi-volatile organic compounds in solid soil matrices. The Polynuclear Aromatic (PNA) and Polychlorinated Biphenyl (PCB) semi-volatiles are studied in this article to determine the feasibility of using this method for the direct thermal extraction and analyses of these compounds from solid soil matrices. Solid soil samples between 1 and 100 milligrams are placed directly into the glass lined stainless steel desorption tube on top of a glass wool plug, and the semi-volatiles are thermally extracted directly into the GC injection port. No solvent extraction or other sample preparation is required. Deuterated internal standards are added to each soil sample for the quantification of the sixteen standard polynuclear aromatic (PNA) compounds specified by EPA method 8275, the polychlorinated biphenyl's (PCB) as well as other semi-volatiles. Accurate identification and quantification of these semi-volatile organic compounds down to the part per billion (ppb) level in less than 100 milligrams of soil are achievable with this technique.
EQUIPMENT
The Short Path Thermal Desorption System Model TD-2 was attached to the injection port of a Hewlett Packard 5890 Series II GC attached to the H.P. 5971 MSD detector. The Short Path Thermal Desorption System and the Direct Thermal Desorption Technique are described in other publications (4, 5, 6 & 7). The data was collected on a PC using the H.P. ChemStation Software. The Mass Spec was operated in the electron impact mode (70 eV) and in both Total Ion and Selected Ion modes, depending on the sample being analyzed and the limits of detection required.
The GC capillary column is a 30 meter x 0.25 mm I.D., 0.5 micron film thickness, DB5-MS Column (J&W Scientific). A short 0.5 meter by 0.53 mm diameter deactivated fused silica guard column was used on the injection port end of the capillary column to act as a cold trapping area for the desorbed materials and also to protect the capillary column and extend its lifetime. The GC was used in the splitless mode of operation and the injection port was maintained at 300 degrees C for the PCB and 350 degrees C for the PNA samples.
For the PNA analyses, the samples were desorbed at 325 degrees C for 20 minutes at a carrier gas flow of 1.5 ml/min into the GC injection port. The semi-volatiles were trapped at the front of the megabore guard column. The GC was maintained at 30 degrees C during the desorption process, after which it was temperature programmed to 200 degrees C at 10 degrees C/min. The oven was subsequently programmed to 250 degrees C at 4 degrees C/min and then to 300 degrees C at 2 degrees C/min for the separation of the 16 PNA's specified in EPA Method 8275. For the identification of the PNA's the mass spec was scanned from 50 to 350 daltons and was used in the SIM mode for the quantification of the PNA samples.
For the PCB analysis the samples were desorbed at 300 degrees C for 10 minutes at a carrier gas flow rate of 1.5 ml/min through the desorption tube. The GC oven was temperature programmed from an initial temperature of 50 degrees C to 200 degrees C at a rate of 10 degrees C/min and then programmed at a rate of 5 degrees C per minute to 300 degrees C. The mass spec was operated in the Selected Ion Mode (SIM) for the identification and quantification of the PCB groups of compounds. The two or three most intense molecular ions for each of the PCB groups of compounds (1 Chlorine through 10 Chlorines) were scanned. The correct isotopic ratios of the second and third most intense ions were used to verify the presence of the PCB's.
The desorption or sampling tubes consist of 1/4" diameter x 4.0" long (3.0 mm I.D.) glass lined stainless steel desorption tubes. The tubes are packed with a small plug of silanized glass wool and then cleaned in the S.I.S. Conditioning Oven at 320 degrees C for 2 to 4 hours with carrier gas flow through the tubes at a rate of 5 to 10 ml/min. This assures that no foreign materials or contamination will interfere with the analysis. Blank tubes were run between each sample to verify this contamination-free environment.
The Short Path Thermal Desorption System Model TD-2, sits directly over the injection port of the Gas Chromatograph (Figure 1). The glass-lined stainless steel desorption tube with glass wool plug and sample is fitted with a short transfer needle and connected to the thermal desorption system. Carrier gas flow is then initiated through the desorption tube for 1.0 minute at a flow rate of 1.5 ml/min to purge it of residual air, volatile solvents and methanol from the matrix spiked internal standard. A pneumatic actuator then drives the desorption tube and needle assembly into the GC injection port. Two temperature regulated heating blocks close around the desorption tube to ballistically heat the samples therein. The heating of the samples in the desorption tube combined with the carrier gas flow through the desorption tube drive any volatiles and semi-volatiles into the GC injection port where they are cryofocused at the front of the GC capillary column. Samples were desorbed for 10 to 20 minutes to effect the complete thermal extraction of the semi-volatiles from the solid matrix soil sample and cryofocus these compounds at the front of the GC capillary guard column. Once the desorption process is complete, the heating blocks are opened, the desorption tube and needle are removed from the GC injection port and the temperature programming of the GC column is commenced to elute the trapped components from the capillary column. The theory and operation of the Short Path Thermal Desorption System have been described in detail in previous publications (4,5,6 & 7).
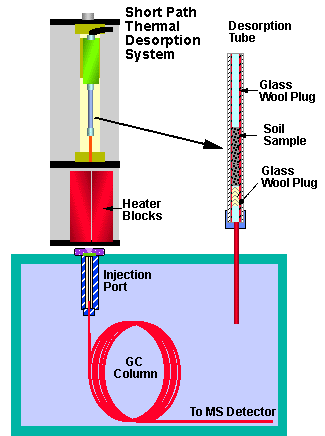
Figure 1 - Theory of Operation - Short Path Thermal Desorption System
EXPERIMENTAL
POLYNUCLEAR AROMATICS (PNA)
For the quantification of the PNA's, a stock solution of the mixture of the 5 deuterated PNA's (d8-naphthalene, d10-acenaphthene, d10-phenanthrene, d12-chrysene, and d12-perylene) was prepared in methanol at a concentration of 27 ng/ul foreach of these internal standards. This solution was labeled as the Internal Standard Stock Solution and was used both for the preparation and dilution of the standard stock solutions of the PNA standards and as a matrix spiked internal standard for the solid soil samples to be quantified. A second stock solution of the 16 standard PNA's (Table I) specified by the EPA methods was prepared by adding the standard 10 PNA compounds to 100 ml of the Internal Standard Stock Solution at a concentration of 100 ng/ul for each of the PNA components. This solution was then consecutively diluted by 50% with the internal standard stock solution to produce concentrations of 100, 50, 25, 12.5, 6.25, 3.12, 1.56, and 0.78 ng/ul of the PNA's in the internal standard solution. The deuterated internal standard concentration remains at 27 ng/ul for each of the samples.
Table I - Quantification of PNA's
Compound Mol.Wt. Ret Time IntStd Slope Intercept Corr. Coef. ------------------- ------- -------- ------ ------- --------- ----------- d8-Naphthalene (I) 136 12.17 Naphthalene 128 12.23 I 0.0313 -0.024 0.999 Acenaphthylene 152 16.28 II 0.0411 -0.012 0.997 d10-Acenaphthlene(II) 164 16.63 Acenaphthene 154 16.71 II 0.0198 0.0018 0.999 Fluorene 166 18.02 II 0.0251 -0.051 0.996 d10-Phenanthrene(III) 188 20.94 Phenenthrene 178 21.01 III 0.0228 -0.024 0.999 Anthracene 178 21.24 III 0.0257 -0.081 0.995 Fluoranthene 202 25.93 III 0.0315 -0.078 0.991 Pyrene 202 27.01 III 0.0321 -0.066 0.993 Benzoanthracene 228 33.98 IV 0.0224 -0.055 0.998 d12-Chrysene (IV) 240 34.05 Chrysene 228 34.31 IV 0.0224 -0.011 0.999 Benzo(b)fluoranthene 252 43.41 V 0.0159 0.076 0.951 Benzo(k)fluoranthene 252 43.56 V 0.0213 -0.023 0.995 Benzo(a)pyrene 252 46.07 V 0.0201 -0.022 0.999 d12-Perylene (V) 264 46.46 Benzo(ghi)perylene 276 55.51 V 0.0174 -0.012 0.908 Dibenzo(a,h)anthracene 278 55.86 V 0.0178 -0.12 0.951 Indeno(1,2,3-cd)pyrene 276 57.11 V 0.0224 -0.12 0.978
In order to generate a calibration curve for the quantification of the PNA's, two (2.0) ml of each of the stock solutions were injected into the desorption tubes on top of the glass wool plug using a 5.0 ml syringe (Figure 1). This provides for 54 ng of the internal standards injected plus PNA amounts ranging from 200 ng down to 1.56 ng. The desorption tubes were attached to the Short Path Thermal Desorption System and a syringe needle was mounted to the bottom of the desorption tube. The Short Path Thermal Desorption system was operated in the automatic mode of operation at a purge flow rate of 1.5 ml/min through the desorption tube. An initial gas purge for 1.0 minute serves to remove the methanol from the inside of the desorption tube. The PNA's remained on the walls of the desorption tube and on the glass wool plug. The desorption tube heater blocks were set to 325 degrees C for the thermal desorption system. After the initial gas purge, the desorption tube needle was injected into the GC injection port, and the desorption system heater blocks were closed around the desorption tube. The semi-volatiles were thermally extacted into the GC injection port for 20 minutes at a flow rate of 1.5 ml/min through the desorption tube (Figure 1). The GC oven remained at 30 degrees C to trap the volatiles purged from the sample in a narrow band at the front of the megabore guard column during the extraction process, after which the column oven was temperature programmed to elute the organics from the column. The oven was programmed to 250 degrees C at 4 degrees C/min and then to 300 degrees C at 2 degrees C/min for the separation of the 16 PNA's and internal standards. For the identification of the PNA's, the mass spec was scanned from 50 to 350 daltons. The mass spec was used in the SIM mode for the quantification of the PNA samples using the most intense ions for each component to generate the calibration curve data.
For the identification and quantification of the PNA's in the solid matrix soil samples, one to 100 milligrams of soil were placed directly into the Glass lined Desorption Tube on top of a glass wool plug. Two ul of internal standard (54 nanograms of d8-naphthalene, d10-acenaphthene, d10-phenanthrene, d12-chrysene, and d12-perylene in methanol) was added on top of the sample. The desorption tube with sample was attached to the S.I.S. Short Path Thermal Desorption System and a syringe needle was attached to the end of the desorption tube. The samples were purged with carrier gas for 1.0 minute to remove the methanol solvent, then injected into the GC and thermally extracted for 20 minutes into the GC injection port at a desorption temperature of 325 degrees C. The semi-volatiles extracted from the soil were trapped at the front of the GC column which was held at 30 degrees C during the thermal extraction process. After the thermal extraction process was complete, the GC oven was temperature programmed at 10 degrees /min to 200 degrees C, then at 4 degrees /min to 250, and finally at 20/min to 300 degrees C to produce the total ion chromatogram. The mass spec (H.P. MSD) detector was scanned from 50 to 350 amu for the complete identification of the semi-volatiles and was used in the selected ion mode for the quantification of the semi-volatile PNA's.
POLYCHLORINATED BIPHENYL'S (PCB'S)
For the quantification of the PCB's, an Internal Standard Stock Solution was prepared by diluting standard phenanthrene in methanol to a concentration of 5.0 ng/ul. This solution was labeled as the Internal Standard Stock Solution and used both for the preparation and dilution of the standard stock solutions of the PCB standards and as a matrix spiked internal standard for the solid soil samples to be quantified. A series of stock solutions of the 9 standard PCB's (Table VI) were diluted with the Internal Standard Stock solution of phenanthrene to prepare standard concentrations of the PCB samples ranging from 50 ng/ul down to 0.1 ng/ul by consecutively diluting the initial PCB solution with the internal standard stock solution.
In order to generate a calibration curve for the quantification of the PCB's, two (2.0) ml of each of the stock solutions were injected into the desorption tubes on top of the glass wool plug using a 5.0 ml syringe (Figure 1). This provides for 10 ng of the internal standards injected plus PCB amounts ranging from 100 ng down to 0.2 ng. The desorption tubes were attached to the Short Path Thermal Desorption System, and a syringe needle was mounted to the bottom of the desorption tube. The Short Path Thermal Desorption system was operated in the automatic mode of operation at a purge flow rate of 1.5 ml/min through the desorption tube. An initial gas purge for 1.0 minute serves to remove the methanol from the inside of the desorption tube. The PCB's remain on the walls of the desorption tube and on the glass wool plug. The desorption tube heater blocks were set to 300 degrees C for the thermal desorption system. After the initial gas purge, the desorption tube was injected into the GC injection port, the desorption system heater blocks were closed around the desorption tube and the semi-volatiles were thermally extacted into the GC injection port for 10 minutes at a flow rate of 1.5 ml/min through the desorption tube (Figure 1). The GC oven remained at 50 degrees C to trap the volatiles purged from the sample in a narrow band at the front of the megabore guard column during the extraction process, after which the column oven was temperature programmed to elute the organics from the column. The oven was programmed from 50 degrees C to 200 degrees C at a rate of 10 degrees C/min and then to 300 degrees C at 5 degrees C/min for the separation of the PCB's and internal standard. The mass spec was used in the SIM mode for the quantification of the PCB samples using the two or three most intense ions for each component to generate the calibration curve data. The second and third most intense ions were used as qualifying ions to verify the presence of PCB's. Accurate isotopic ratios must be present in order to quantify the various PCB's.
The PCB soil samples were analyzed and quantified much the same as the PNA compounds above. Soil samples from 1.0 to 100 milligrams are weighed and transferred to the desorption tube and placed on top of the glass wool plug (Figure 1) Two ul of the Internal standard were injected onto the GLT desorption tube on top of the soil sample. The Samples were desorbed at 300 degrees C for 10 minutes with a carrier gas flow of 1.5 ml/min through the desorption tube. The GC column was kept at 50 degrees C during the desorption after which it was temperature programmed. The mass spec was operated in the SIM mode using the major ions for the PCB's plus two qualifier ions of the next most intensity (Table V).
RESULTS AND DISCUSSION
POLYNUCLEAR AROMATICS (PNA'S)
A series of studies were conducted to determine the optimum conditions for desorbing the 16 PNA's specified in EPA methods for PNA's in environmental samples. Standards of PNA's were desorbed at temperatures of 250, 275, 300, 325, 350, 375 and 400 degrees C in order to determine the optimum temperature for the detection of all the 16 PNA's. Temperatures at 300 degrees C and lower were determined to be insufficient for the detection of all the PNA's. Desorption temperatures of 325 to 350 degrees C were determined to be the optimum temperatures for the thermal desorption of the PNA's. Temperatures above 350 degrees C were found to be quickly detrimental to the GC guard column, when actual sand and soil samples were analyzed. The compounds which came off the standard sands and soil at these elevated temperatures contaminated the front of the guard column, resulting in loss of sensitivity of detection and loss of column resolution. The problem was quickly solved by replacing the 0.5 meter megabore deactivated guard column to restore the column to its original performance. The desorption times were varied from 5 minutes up to 60 minutes at a desorption temperature of 325 degrees C and a desorption tube flow of 1.5 ml/min in order to determine the optimum desorption conditions. The optimum time of desorption was determined to be 10 to 20 minutes. Beyond that no additional sensitivity was apparent. Based on these studies, the desorption conditions were set to a 325 degrees C desorption temperature for 20 minutes at a flow of 1.5 ml/min of carrier gas through the desorption tube. A chromatogram of the 16 PNA's and 5 internal standards used in this method is shown in Figure 2, and the PNA compounds and internal standards are listed in Table I. With the exception of Benz[b]fluoranthene and Benzo[k]fluoranthene [Compounds (15 & 16)], all the compounds were baseline resolved when analyzed using the most intense ions for each of the PNA's in the selected ion profiles of the chromatogram.
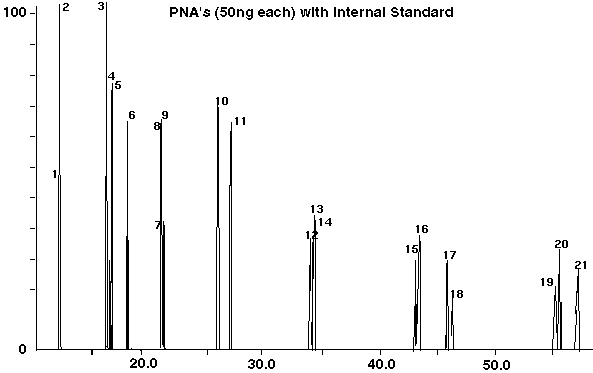
Figure 2 - Chromatogram of PNA Standards
QUANTIFICATION OF PNA'S
The peak area integrations of the highest mass molecular ions for each of the PNA's were used to generate the calibration curves for each of the PNA's relative to the closest internal standard. The curves were constructed by plotting the ratio of the PNA area / the area of the closest internal standard (y-axis) versus the concentration of the PNA (x axis). A typical curve is shown in Figure 3, and the data for all the compounds is displayed in Table I. A linear calibration curve was obtained for all the PNA standards from concentrations ranging from 100 nanograms down to 0.4 ng. Regression analysis of the curves follows the formula:
y = a * x + b
Where y = the Area of the PNA/Area of the Internal Standard
x = The Concentration of the PNA in Nanograms
a = The Slope of the Line in the Chart for the PNA
And B = The y Axis Intercept of the Calibration Curve
Solving this equation for x (the concentration of the PNA) gives:
x = (y - b) / a
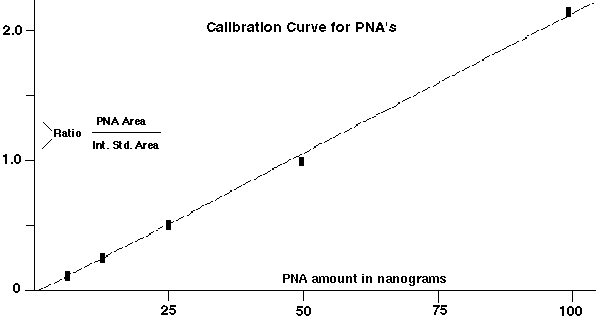
Figure 3 - Calibration Curve For Polynuclear Aromatics
In this method, soil samples can be analyzed ranging from 1.0 mg up to 100 mg. The limits of sample size are due to the accuracy of weighing samples less than 1.0 milligram and the maximum capacity of the desorption tube (approx. 500 mg). Another concern in analyzing small samples is the homogeniality of sample. Small samples must be thoroughly ground up and mixed to assure a good representative sampling for the soil samples. Higher concentrations of PNA's in soil samples and better homogeniality of soil samples could be achieved by using larger soil samples (100 to 500 mg) and the GC injection port in the split mode of operation as so not to overload the MS detector. Therefore, it is possible to achieve quantification of PNA's in soil at levels from 10 ppb to 100 ppm using this technique in sample sizes ranging from 1.0 milligram to 100 milligram as documented in Table II. The data in Table II is based on the minimum and maximum sensitivities of the H.P. 5971 MSD for the PNA compounds analyzed under normal operating conditions with signal to noise ratios of at least 10:1. Newer mass spectrometers with higher sensitivities could increase this sensitivity 10 to 100 fold.
Table II - Detection Levels of PNA's in Soil Samples
Soil Sample size Min detection level Max. detection level ------------------ ------------------- -------------------- MSD Detection Limit 1 ng 100 ng 1.0 milligrams 1 ppm 100 ppm 10.0 milligrams 100 ppm 10 ppm 100.0 milligrams 10 ppb 1 ppm
A soil sample from a super fund dump site was analyzed by placing 10.0 milligrams into the desorption tube along with the internal standard. The resultant total ion chromatogram is shown in Figure 4 and the selected ion chromatogram is shown in Figure 5. The data on the analysis of the super fund dump site soil is displayed in Table III. Figure 4 is a total ion chromatogram, scanning the mass range from 50 to 350 daltons. This data permitted a total mass spec search of all the components with the identification of most of the peaks as displayed in Table III. Additional semi-volatile compounds were also identified in this sample beyond the 16 standard PNA's (Table III, Left Column).
Table III - Analysis of Soil From Superfund Dump Site
No. Compound No. Polynuclear Aromatics Amount(ppm) -- ----------------------------- --- --------------------- ----------- A. Methyl Naphthalene 2. Naphthalene 17 B. 1,2-Isobenzofurandione 3. Acenaphthylene 66 C. Dibenzofuran 5. Fluorene 06 D. 9H-Fluoren-9-one 6. Fluorene 11 E. Naphthanene-2,3-dicarboxylic 8. Phenanthrene 20 F. 9-Methyl-Phenanthrene 9. Anthracene 165 G. 2-Methyl-Phenthrene 10. Fluoranthene 100 H. 9-Methyl-Anthracene 11. Pyrene 147 I. 9,10-Anthracenedione 12. Benzoanthracene 14 J. 1-Methyl-Pyrene 14. Chrysene 266 K. Benzo(b)fluorene 15. Benzo(b)fluoranthene 187 L. 2-Methyl-Pyrene 16. Benzo(k)fluoranthene 129 M. 4-Methyl-Pyrene 17. Benzo(a)pyrene 186 N. Benzo(b)naptho(2,1-d)thiophene 19. Benzo(ghi)perylene 4 O. Cyclopenta(cd)pyrene 20. Dibenzo(a,h)anthracene 4 P. Benzo(b)naptho(2,1-d)thiophene 21. Indendo(1,2,3-cd)Pyrene 17 Q. Triphenylene R. 9-Methyl-Benz(a)anthracene S. 1-Methylchrysene T. 3-Methylchrysene U. 2,2'-Binaphthalene V. Benzo(i)fluorene W. Perylene
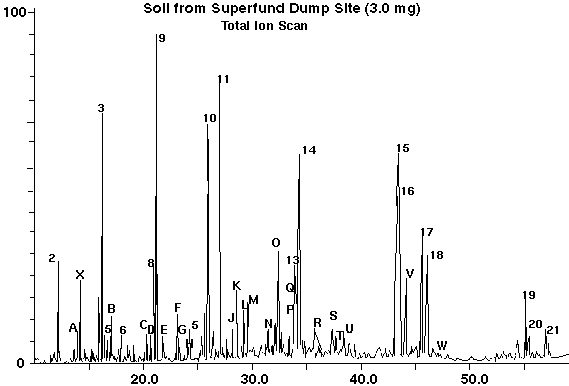
Figure 4 - Total Ion Scan of Superfund Dump Site Soil
In Figure 5, the soil sample was analyzed in the SIM mode described previously to simplify the chromatogram and display the peaks for the particular PNA's to be quantified. The quantitative date for the PNA's is displayed in Table III, Right Column.
A study was conducted by spiking clean sand with the PNA mix at a concentration of 10.0 ppm (10.0 ng of each of the PNA's per 1.0 mg of sand). As described previously, the samples were spiked with the PNA's in methanol such that the entire soil was wet. The sample was allowed to stand for several hours and then the methanol was evaporated off the sand by heating it slightly to 40 degrees C and flushing the sand with a stream of dry nitrogen. This assured that the PNA's were thoroughly and evenly distributed through the sand sample; however, some sample loss was anticipated in this evaporation process, especially of the low boilers. The chromatogram of a 10.0 milligram sample of the sand is shown in Figure 6. Ten samples of the sand were analyzed. The data consisting of the average amount of PNA detected and the standard deviations are listed in Table IV.
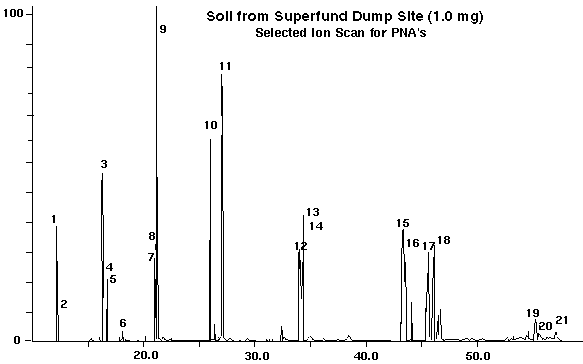
Figure 5 - SIM scan for 16 PNA's in Superfund Dump Site Soil
Table IV - Quantification of PNA's in Spiked Sand Samples (10 ppm)
No. Polynuclear Aromatic PNA amount (ppm) Standard Deviation --- --------------------- ---------------- ------------------- 2 Naphthalene 6.0 2.0 3 Acenaphthylene 5.1 0.3 5 Acenaphthene 6.5 2.7 6 Fluorene 7.8 2.5 8 Phenanthrene 8.5 3.9 9 Anthracene 4.7 1.9 10 Fluoranthene 5.6 2.7 11 Benzoanthracene 4.8 2.3 12 Benzoanthracene 7.2 0.5 14 Chrysene 9.5 1.2 15 Benzo[k]fluoranthene 6.0 1.0 16 Benzo[k]fluoranthene 5.6 0.8 17 Benzo[a]pyrene 5.2 1.1 19 Benzo[gho]perylene 8.3 2.8 20 Benzo[a,h}anthracene 9.4 3.8 21 Indeno[1,2,3-cd]pyrene 7.0 2.2
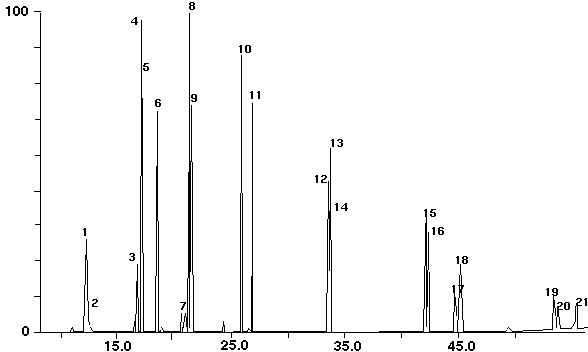
Figure 6 - Sand Sample Spiked with 10 ppm of 16 PNA's
The PNA's were quantified at levels between 47% and 95% of the spiked levels. Errors could have resulted due to (a) the methanol had not completely evaporated resulting in a sample actually weighing less than weighed out, (b) accuracy of weighing 10.0 milligram samples, (c) loss of PNA due to inefficient thermal extraction in the direct thermal extraction method, (d) binding effects of the PNA's to the soils and (e) loss of PNA concentration due to evaporation or loss due to PNA's adhering to the vessel used to mix and evaporate the PNA's from the soil. This last source of error of sample loss due to PNA's adhering to the sides of the sample preparation vessel is undoubtedly the largest contributor to PNA loss. In the sample preparation, 10 grams of soil with 3.0 ml of methanol were evaporated in a 25 ml Erlenmeyer flask. This flask exposed the sample to a large surface area for the possible adhesion of the PNA's. The loss of sample could also be due to inefficient thermal extraction of the PNA's from the soil. However, it is apparent that this loss is minimal, since the entire boiling point range of PNA's exhibited similar yields and the highest boiling compounds still gave good yields. Studies in which the PNA's were spiked on top of sand samples in the desorption tubes produced yields of better than 90%, confirming some of these possible sources of sample loss. Future studies will investigate these sources of error in sample preparation.
POLYCHLORINATED BIPHENYL'S (PCB'S)
A study was undertaken similar to the study for the PNA's above to determine the optimum conditions for the direct thermal desorption of the PCB's from solid matrix samples. From these studies, it was determined that a desorption temperature of 300 degrees C for 10 minutes with a carrier gas flow through the desorption tube of 1.5 ml/min was sufficient for the extraction of the entire range of PCB's. For this study, a 0.5 meter blank deactivated fused silica column was placed into a GC with a FID detector. The Short Path Thermal Desorption System was attached to the GC injection port. A desorption tube spiked with the PCB standard was injected into the GC injection port, and the heated desorption tube blocks were closed around the desorption tube. The FID detector detected the entire range of semi-volatiles, as they were desorbed from the desorption tube. The resultant chromatogram of an Aroclor 1268 sample analyzed in this manner is shown in Figure 7. The Extraction ion profile (Figure 7) provides proof that within 5 minutes all of the PCB's have been thermally extracted from the sample. For 100% assurance that the extraction is complete, a desorption time of 10.0 minutes is used for all PCB suspected samples.
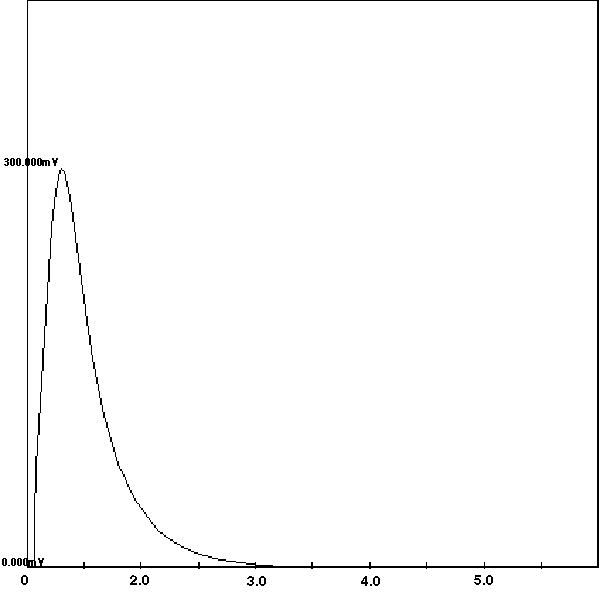
Figure 7 - Extraction Profile of PCB's From a Spiked Desorption Tube
Table V - Mass Spec Ions of the PCB family of Compounds
No.Cl Mol Wt M M+2 M+4 M+6 M+8 M+10
----- ----- -------- -------- -------- -------- ------- -------
0 154 154(100) 156(0.8)
1 188 188(100) 190(33)
2 222 222(100) 224(65) 226(11)
3 256 256(100) 258(97) 260(32) 262(4)
4 290 290(75) 292(100) 294(49) 296(11) 298(1)
5 324 324(60) 326(100) 328(64) 330(31) 332(3) 334(0.2)
6 358 358(50) 360(100) 362(80) 364(35) 366(8) 368(1)
7 392 392(43) 394(100) 396(80) 398(52) 400(17) 402(3)
8 426 426(32) 428(89) 430(100) 432(64) 434(26) 436(7)
9 460 460(27) 462(78) 464(100) 466(75) 468(36) 470(12)
10 494 494(22) 496(69) 498(100) 500(86) 502(48) 504(19)
SIM Ions in Bold (xxc) - relative intensity of ions
For the analysis of the PCB's, the mass spec was used in the selected ion mode (SIM) due to the higher sensitivities available with this mode and the ability to simplify the chromatograms by selectively analyzing only the chlorinated compounds of interest. Since the PCB's are all chlorinated, the isotope ions and their ratios of intensity are useful for both the detection and the confirmation of the PCB compounds. The data for all the groups of PCB's is presented in Table V.
For the detection and analyses of the various isomer groups of the PCB's, the most intense ion and the next most two or three ions were used by the mass spec in the SIM mode to search for these compounds and then eventually to quantify the PCB's in various soil samples. A variety of PCB standards were analyzed by the method developed to demonstrate its use. The Chromatogram of an Aroclor mix of all the PCB's is shown in Figure 8. The numbers on the chromatogram indicate the number of chlorines identified in the peak. It is evident that all the Biphenyl's from plain Biphenyl (0 Chlorines) through DecachloroBiphenyl (10 chlorines) were detectable. Additional Aroclors 1221 (Figure 9), 1242 (Figure 10), 1254 (Figure 11) and 1268 (Figure 12) indicated the changing characteristic patterns for the various aroclors. In particular the Aroclor 1268 has very high percentages of the 9-ChlorineBiphenyls and Decachlorobiphenyl.
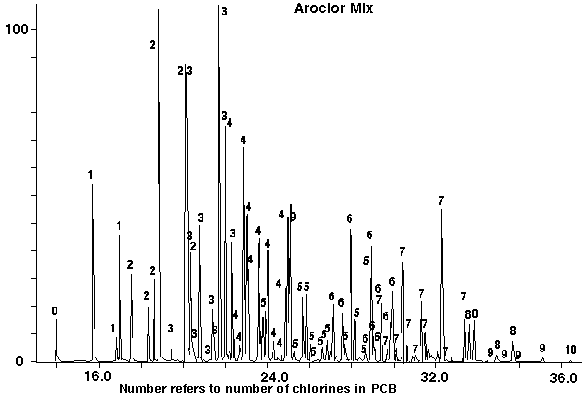
Figure 8 - Standard PCB Aroclor Mix
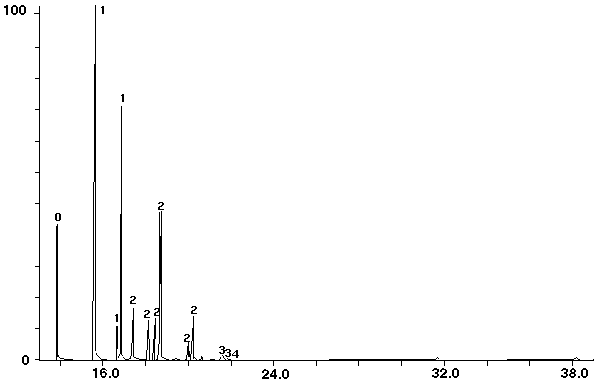
Figure 9 - Aroclor 1221 Standard
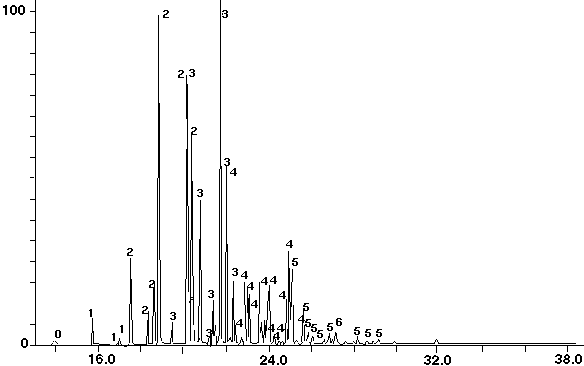
Figure 10 - Aroclor 1242 Standard
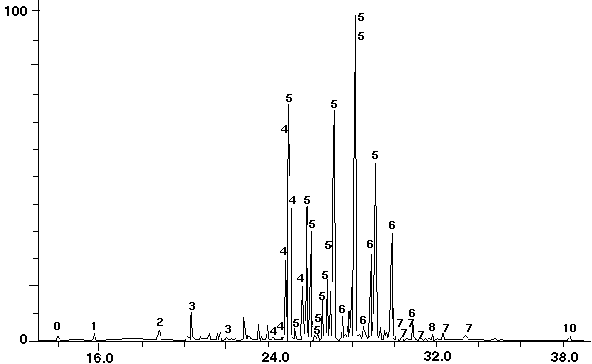
Figure 11 - Aroclor 1254 Standard
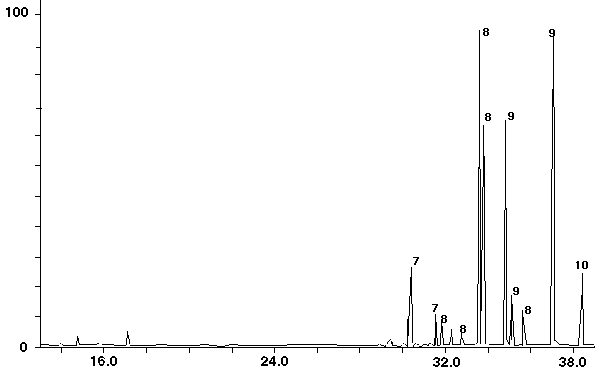
Figure 12 - Aroclor 1268 Standard
Standard methods for the quantification of the PCB's in environmental samples quantitate these samples by identifying the PCB patterns (see Figures 9-12) of the class of PCB compounds and then quantifying by summing the total count of the chlorinated compounds. This method appears to be inaccurate, especially when mixtures of Aroclors are involved. We are in the process of proposing a more accurate identification and quantification of the individual PCB's in the soil samples. Although this may be more time-consuming and require the use of the mass spec using SIM methods, the results are much more accurate. For these initial studies, a mix of 9 common PCB compounds and the compound Phenanthrene were used as the internal standard. For a true analytical method, a much larger number of PCB's would need to be analyzed and calibration curves established. The compounds used for this initial study are listed in Table VI and their chromatogram is shown in Figure 13. For the quantification, the most intense ion for each of the PCB's was used, but the next most two intense ions were also used as qualifying ions. The proper ratio of the qualifier ions to the most intense ion must fall within strict error limits in order for the PCB compound to be quantified. This provides for an extremely accurate identification and quantification of the PCB compounds. The peak area integration of the most intense ions for each of the PCB compounds was used to generate the calibration curves for each of the PCB's relative to integrated peak areas of the Phenanthrene internal standard. The curves were constructed by plotting the PCB area / the area of the Phenanthrene internal standard (y axis) versus the concentration of the PCB (x axis). A typical calibration curve is shown in Figure 14 and the data for the curves is shown in Table VI. A linear calibration curve was obtained for all the PCB's analyzed at concentrations ranging from 0.1 nanograms to 100 nanograms. Regression analysis of the curves produced the formula:
y = a * x + b
Where y = The Area of the PCB / Area of the Internal Standard Phnanthrene
x = The Concentration of the PCB in Nanograms
a = The Slope of the Line in the Chart for the PCB
And b = The y Axis Intercept of the Calibration Curve
Solving this equation for X (the concentration of the PCB) gives:
x = (y - b) / a
Table VI - Quantification of PCB's in a Contaminated Dump Site Soil
# No. Cl Biphenyl Molec. Wt. Ret.Time EPA Soil(ppm) -- ------ ---------------------- ----------- ---------- -------------- A. 1 2-Mono 188 16.69 3.93 B. 2 2,3-Di- 222 18.77 0.75 C. 3 2,4,5-Tri- 256 21.12 1.12 D. 4 2,2',4,6-Tetra- 290 21.52 0.82 E. 5 2,3',3,4,5'-Penta- 358 26.75 3.55 F. 6 2,2',4,4',5,6'-Hexa- 358 27.02 3.26 G. 7 2,2',3,4',5,6,6'-Hepta 392 28.48 7.98 H 8 2,2',3,3',4',5,6,6'-Octa- 426 31.77 2.55 I. 10 Deca- 494 28.32 2.41
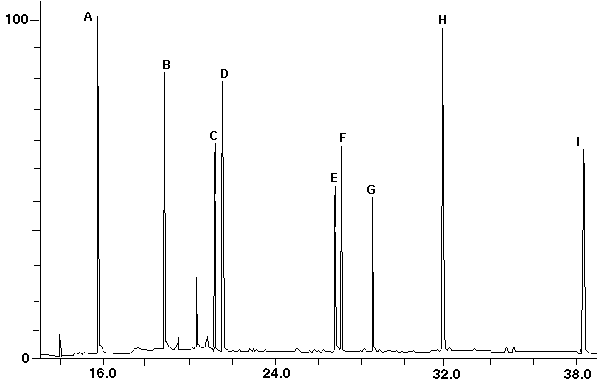
Figure 13 - Total Ion Chromatogram of PCB Standard Mix
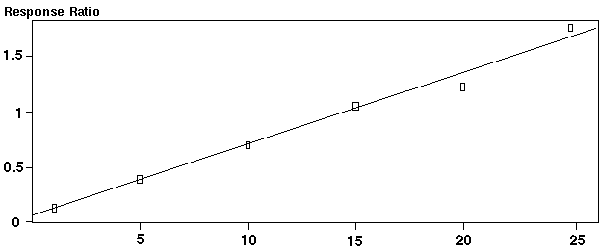
Figure 14 - Calibration Curve For PCB's
In this method, soil samples can be analyzed ranging from 1.0 mg up to 100 mg. The limits of sample size are due to the accuracy of weighing samples less than 1.0 milligram and the maximum capacity of the desorption tube (approx. 500 mg). Another concern in analyzing small samples is the homogeniality of sample. Small samples must be thoroughly ground up and mixed to assure a good representative sampling for the soil samples. Higher concentration of PCB's and greater homogeniality of soil samples could be achieved by using larger soil samples (100 to 500 mg) with the GC injection port in the split mode and splitting the sample 10:1. This would avoid overloading of the mass spectrometer detector. Therefore, it is possible to achieve quantification of PCB's in soil at levels from 1.0 ppb to 100 ppm in sample sizes ranging from 1.0 milligram to 100 milligram using this technique, as documented in Table VII.
Table VII - Detection Levels of PCB's in Soil Samples
Soil Sample size Min detection level Max. detection level -------------------- ------------------------- -------------------- MSD Detection Limit 1 ng 100 ng 1.0 milligrams 1 ppm 100 ppm 10.0 milligrams 100 ppm 10 ppm 100.0 milligrams 10 ppb 1 ppm
Figure 15 is the chromatogram for the analysis of a soil sample from a hazardous waste dump site. It was analyzed using the method described above. The key PCB's in this sample were quantified. The results are listed in Table VI.
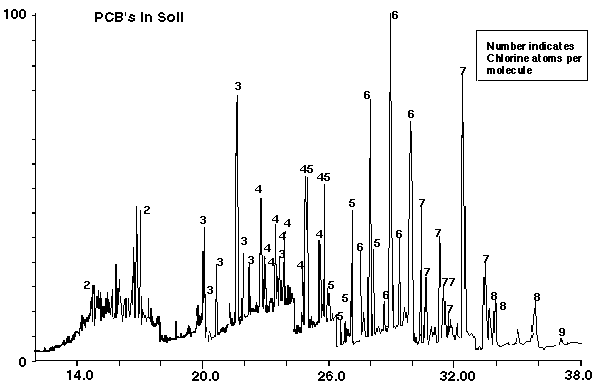
Figure 15 - Contaminated Dump Site Soil
CONCLUSION
The Direct Thermal Desorption technique utilizing the Short Path Thermal Desorption System has proven to be a versatile and time-saving technique for both the qualitative and quantitative analysis of the both the volatiles and semi-volatiles in soil samples. Utilizing this technique, no solvent extraction or other sample preparation is required. This eliminates the problem of generating additional hazardous wastes during the analysis of these hazardous waste samples. Solid soil samples between 1 and 100 milligrams are placed directly into the GLT desorption sampling tube without any sample preparation. Deuterated internal standards are added for the quantification of the PNA's and PCB's in the soil samples. The semi-volatiles are then thermally extracted directly from the sample into the GC injection port for subsequent analysis via GC/MS techniques to identify and quantify the semi-volatiles. PNA's, PCB's and other semi-volatiles can be detected and quantified in soils from 1 ppb to 100 ppm with a high degree of accuracy and reliability. The technique can be readily modified for the direct identification and quantification of other volatiles and semi-volatiles in solid matrix samples without the need for sample extraction or other sample preparation. This provides for a quick, efficient and accurate method of analysis for a wide range of volatiles and semi-volatiles in solid samples without any sample preparation.
REFERENCES
Manura, J.J., Hartman, T.G., Applications of a Thermal Desorption GC Accessory, American Laboratory,
May, 1992, 46-52 (1992).
Manura, J.J., Direct Thermal Analysis of Plastic Food Wraps, The Mass Spec Source, Vol. XIV (2), 14-20
(1991).
Manura, J.J., Direct Thermal Analysis Using the Short Path Thermal Desorption System: A New Technique to
Permit he Analysis of Volatiles and Semi-volatiles in Solid Samples without Solvent Extraction, The Mass
Spec Source, Vol. XII (2), 22-27 (1991)
Manura, J.J., Overton, S.V., Baker, C.W., and Manos, J.N., Short Path Thermal Desorption - Design and
Theory, The Mass Spec Source, Vol. XIII (4), 22-28, (1990).
Quantification of Naphthalene in a Contaminated Pharmaceutical Product by Short Path Thermal Desorption,
S.I.S. Application Note No. 12, May, 1992.
Robbat, A, Tyng-Yun Liu & Abraham, B., Evaluation of a Thermal Desorption Gas Chromatograph/Mass
Spectrometer: On-Site Detection of Polychlorinated Biphenyls at a Hazardous Waste Site, Anal. Chem., 64,
358-364 (1992).
Robbat, A., Tyng-Yun Liu & Abraham, B., On-Site Detection of Polycyclic Aromatic Hydrocarbons in
Contaminated Soils by Thermal Desorption Gas Chromatography/Mass Spectrometry, Anal. Chem, 64,
1477-1483 (1992).
Snyder, J., Grob, R., McNally, M., & Oostdyk, T., Comparison of Supercritical Fluid Extraction With
Classical Sonication and Soxhlet Extractions for Selected Pesticides, Anal.Chem., 64, 1940-1946 (1992).

