- ▶
- Heaters/Source
- ▶
- Agilent Heaters and SensorsMass Spectrometry, Scientific Supplies & ManufacturingScientific Instrument Services 5973 Source Heater Tamper Resistant Allen Wrench 5973/5975 Quad Sensor 5985 Source Heater Assembly Agilent Interface Heater Assembly 5971 Interface Heater

- ▶
- Reference Material on InstrumentationArticle - A High Temperature Direct Probe for a Mass Spectrometer Design of a Direct Exposure Probe and Controller for use ona Hewlett-Packard 5989 Mass Spectrometer SIS AP1000 AutoProbe™ SIS AP2000 AutoProbe™ - Description of System HPP7: Direct Probe Electronics Console HPP7: Direct Probe for the Agilent (HP) 5973/5975 MSD HPP7: HP Direct Probe Application Notes HPP7: Installation Directions for the Direct Probe HPP7: Side Cover for the HP 5973 MSD HPP7: Support HPP7: Probe Inlet System for the Agilent (HP) 5973 and 5975 MSD with Automatic Indexed Stops HPP7: Theory of Operation of the Direct Probe and Probe Inlet System Direct Thermal Extraction Thermal Desorption Application Notes Environmental Thermal Desorption Application Notes Food Science Thermal Desorption Application Notes Forensic Thermal Desorption Application Notes GC Cryo-Trap Application Notes Headspace Application Notes Purge & Trap Thermal Desorption Application Notes Theory of Operation of the AutoDesorb® System AutoDesorb Notes for SIS Dealers Adsorbent Resin Application Notes Installation of the Short Path Thermal Desorption System on Agilent (HP) and Other GCs Installation of the Short Path Thermal Desorption System on a Varian 3400 GC AutoDesorb® System Development Team Thermal Desorption Applications and Reference Materials Installation of the Short Path Thermal Desorption System - TD5 Part I - Design & Operation of the Short Path ThermalDesorption System Installation Instructions for the Model 951 GC Cryo-Trap on the HP 5890 Series GC Installation Instructions for the Model 961 GC Cryo-Trap on the HP 5890 Series GC Operation of the Model 951/961 GC Cryo-Trap SIS GC Cryo Traps - Theory of Operation NIST/EPA/NIH Mass Spectral Enhancements - 1998 version (NIST98) SIMION 3D Ion Optics Class Mass Spectrometer Source Cleaning Methods MS Tip: Mass Spectrometer Source Cleaning Procedures Mass Spec Source Cleaning Procedures Micro-Mesh® Abrasive Sheets Research Papers Using New Era Syringe Pump Systems EI Positive Ion Spectra for Perfluorokerosene (PFK) Cap Liner Information How do I convert between fluid oz and milliliters? Which bottle material should I choose? Which bottle mouth should I choose? The Bottle Selection Guide CGA Connections for Gas Tanks Chemical Reaction Interface Mass Spectrometry (CRIMS)

- LiteratureApplication Notes Adsorbent Resins Guide Mass Spec Tips SDS Sheets FAQ MS Calibration Compound Spectra Manuals MS Links/Labs/ Organizations MS Online Tools Flyers on Products/Services Scientific Supplies Catalog About Us NextAdvance Bullet Blender® Homogenizer Protocols Micro-Mesh® Literature Instrumentation Literature Agilent GC/MS Literature SIS News / E-Mail Newsletter NIST MS Database - Update Notifications

- ▶
- Chemical Reaction Interface Mass Spectrometry (CRIMS)51. Development and Characterization of a New Chemical Interface for the Detection of Nonradioisotopically Labeled Analytes Using Mass Spectrometry (CRIMS) (EAS 96) 67. Using Chemical Reaction Interface (CRIMS) to Monitor Bacteria Transport in Situ (PittCon 98) 68. Using a Plug In UV-Vis Spectrometer to Monitor the Plasma Conditions in a GC CRIMS (EAS 98) 76. Determination of the Sensitivity of a CRIMS System (ASMS 98)

- Application NotesNote 103: EPA Method 325B, Novel Thermal Desorption Instrument Modification to Improve Sensitivity Note 102: Identification of Contaminants in Powdered Beverages by Direct Extraction Thermal Desorption GC/MS Note 101: Identification of Contaminants in Powdered Foods by Direct Extraction Thermal Desorption GC/MS Note 100: Volatile and Semi-Volatile Profile Comparison of Whole Versus Cracked Versus Dry Homogenized Barley Grains by Direct Thermal Extraction Note 99: Volatile and Semi-Volatile Profile Comparison of Whole vs. Dry Homogenized Wheat, Rye and Barley Grains by Direct Thermal Extraction GC/MS Note 98: Flavor and Aroma Profiles of Truffle Oils by Thermal Desorption GC/MS Note 97: Flavor Profiles of Imported and Domestic Beers by Purge & Trap Thermal Desorption GC/MS Note 96: Reducing Warping in Mass Spectrometer Filaments, with SISAlloy® Yttria/Rhenium Filaments Note 95: Detection of Explosives on Clothing Material by Direct and AirSampling Thermal Desorption GC/MS Note 94: Detection of Nepetalactone in the Nepeta Cataria Plant by Thermal Desorption GC/MS Note 93: Detection of Benzene in Carbonated Beverages with Purge & Trap Thermal Desorption GC/MS Note 92: Yttria Coated Mass Spectrometer Filaments Note 91: AutoProbe DEP Probe Tip Temperatures Note 90: An Automated MS Direct Probe for use in an Open Access Environment Note 89: Quantitation of Organics via a Mass Spectrometer Automated Direct Probe Note 88: Analysis of Silicone Contaminants on Electronic Components by Thermal Desorption GC-MS Note 87: Design and Development of an Automated Direct Probe for a Mass Spectrometer Note 86: Simulation of a Unique Cylindrical Quadrupole Mass Analyzer Using SIMION 7.0. Note 85: Replacing an Electron Multiplier in the Agilent (HP) 5973 MSD Note 84: Vacuum Pump Exhaust Filters - Charcoal Exhaust Traps Note 83: Vacuum Pump Exhaust Filters - Oil Mist Eliminators Note 82: Vacuum Pump Exhaust Filters Note 81: Rapid Bacterial Chemotaxonomy By DirectProbe/MSD Note 80: Design, Development and Testing of a Microprocessor ControlledAutomated Short Path Thermal Desorption Apparatus Note 79: Volatile Organic Compounds From Electron Beam Cured and Partially Electron Beam Cured Packaging Using Automated Short Path Thermal Desorption Note 78: A New Solution to Eliminate MS Down-Time With No-Tool-Changing of Analytical GC Columns Note 77: The Determination of Volatile Organic Compounds in VacuumSystem Components Note 76: Determination of the Sensitivity of a CRIMS System Note 75: An Apparatus for Sampling Volatile Organics From LivePlant Material Using Short Path Thermal Desorption Note 74: Examination of Source Design in Electrospray-TOF Using SIMION 3D Note 73: The Analysis of Perfumes and their Effect on Indoor Air Pollution Note 72: 1998 Version of the NIST/EPA/NIH Mass Spectral Library, NIST98 Note 71: Flavor Profile Determination of Rice Samples Using Shor tPath Thermal Desorption GC Methods Note 70: Application of SIMION 6.0 To a Study of the Finkelstein Ion Source: Part II Note 69: Application of SIMION 6.0 To a Study of the Finkelstein Ion Source: Part 1 Note 68: Use of a PC Plug-In UV-Vis Spectrometer To Monitor the Plasma Conditions In GC-CRIMS Note 67: Using Chemical Reaction Interface Mass Spectrometry (CRIMS) To Monitor Bacterial Transport In In Situ Bioremediation Note 66: Probe Tip Design For the Optimization of Direct Insertion Probe Performance Note 65: Determination of Ethylene by Adsorbent Trapping and Thermal Desorption - Gas Chromatography Note 64: Comparison of Various GC/MS Techniques For the Analysis of Black Pepper (Piper Nigrum) Note 63: Determination of Volatile and Semi-Volatile Organics in Printer Toners Using Thermal Desorption GC Techniques Note 62: Analysis of Polymer Samples Using a Direct Insertion Probe and EI Ionization Note 61: Analysis of Sugars Via a New DEP Probe Tip For Use With theDirect Probe On the HP5973 MSD Note 60: Programmable Temperature Ramping of Samples Analyzed ViaDirect Thermal Extraction GC/MS Note 59: Computer Modeling of a TOF Reflectron With Gridless Reflector Using SIMION 3D Note 58: Direct Probe Analysis and Identification of Multicomponent Pharmaceutical Samples via Electron Impact MS Note 57: Aroma Profiles of Lavandula species Note 56: Mass Spec Maintenance & Cleaning Utilizing Micro-Mesh® Abrasive Sheets Note 55: Seasonal Variation in Flower Volatiles Note 54: Identification of Volatile Organic Compounds in Office Products Note 53: SIMION 3D v6.0 Ion Optics Simulation Software Note 52: Computer Modeling of Ion Optics in Time-of-Flight mass Spectrometry Using SIMION 3D Note 51: Development and Characterization of a New Chemical Reaction Interface for the Detection of Nonradioisotopically Labeled Analytes Using Mass Spectrometry (CRIMS) Note 50: The Analysis of Multiple Component Drug Samples Using a Direct Probe Interfaced to the HP 5973 MSD Note 49: Analysis of Cocaine Utilizing a New Direct Insertion Probe on a Hewlett Packard 5973 MSD Note 48: Demonstration of Sensitivity Levels For the Detection of Caffeine Using a New Direct Probe and Inlet for the HP 5973 MSD Note 47: The Application Of SIMION 6.0 To Problems In Time-of-Flight Mass Spectrometry Note 46: Delayed Extraction and Laser Desorption: Time-lag Focusing and Beyond Note 45: Application of SIMION 6.0 to Filament Design for Mass Spectrometer Ionization Sources Note 44: The Design Of a New Direct Probe Inlet For a Mass Spectrometer Note 43: Volatile Organic Composition In Blueberries Note 42: The Influence of Pump Oil Purity on Roughing Pumps Note 41: Hydrocarbon Production in Pine by Direct Thermal Extraction Note 40: Comparison of Septa by Direct Thermal Extraction Note 39: Comparison of Sensitivity Of Headspace GC, Purge and Trap Thermal Desorption and Direct Thermal Extraction Techniques For Volatile Organics Note 38: A New Micro Cryo-Trap For Trapping Of Volatiles At the Front Of a GC Capillary Column Note 37: Volatile Organic Emissions from Automobile Tires Note 36: Identification Of Volatile Organic Compounds In a New Automobile Note 35: Volatile Organics Composition of Cranberries Note 34: Selection Of Thermal Desorption and Cryo-Trap Parameters In the Analysis Of Teas Note 33: Changes in Volatile Organic Composition in Milk Over Time Note 32: Selection and Use of Adsorbent Resins for Purge and Trap Thermal Desorption Applications Note 31: Volatile Organic Composition in Several Cultivars of Peaches Note 30: Comparison Of Cooking Oils By Direct Thermal Extraction and Purge and Trap GC/MS Note 29: Analysis Of Volatile Organics In Oil Base Paints By Automated Headspace Sampling and GC Cryo-Focusing Note 28: Analysis Of Volatile Organics In Latex Paints By Automated Headspace Sampling and GC Cryo-Focusing Note 27: Analysis of Volatile Organics In Soils By Automated Headspace GC Note 26: Volatile Organics Present in Recycled Air Aboard a Commercial Airliner Note 25: Flavor and Aroma in Natural Bee Honey Note 24: Selection of GC Guard Columns For Use With the GC Cryo-Trap Note 23: Frangrance Qualities in Colognes Note 22: Comparison Of Volatile Compounds In Latex Paints Note 21: Detection and Identification Of Volatile and Semi-Volatile Organics In Synthetic Polymers Used In Food and Pharmaceutical Packaging Note 20: Using Direct Thermal Desorption to Assess the Potential Pool of Styrene and 4-Phenylcyclohexene In Latex-Backed Carpets Note 19: A New Programmable Cryo-Cooling/Heating Trap for the Cryo-Focusing of Volatiles and Semi-Volatiles at the Head of GC Capillary Columns Note 18: Determination of Volatile Organic Compounds In Mushrooms Note 17: Identification of Volatile Organics in Wines Over Time Note 16: Analysis of Indoor Air and Sources of Indoor Air Contamination by Thermal Desorption Note 14: Identification of Volatiles and Semi-Volatiles In Carbonated Colas Note 13: Identification and Quantification of Semi-Volatiles In Soil Using Direct Thermal Desorption Note 12: Identification of the Volatile and Semi-Volatile Organics In Chewing Gums By Direct Thermal Desorption Note 11: Flavor/Fragrance Profiles of Instant and Ground Coffees By Short Path Thermal Desorption Note 10: Quantification of Naphthalene In a Contaminated Pharmaceutical Product By Short Path Thermal Desorption Note 9: Methodologies For the Quantification Of Purge and Trap Thermal Desorption and Direct Thermal Desorption Analyses Note 8: Detection of Volatile Organic Compounds In Liquids Utilizing the Short Path Thermal Desorption System Note 7: Chemical Residue Analysis of Pharmaceuticals Using The Short Path Thermal Desorption System Note 6: Direct Thermal Analysis of Plastic Food Wraps Using the Short Path Thermal Desorption System Note 5: Direct Thermal Analysis Using the Short Path Thermal Desorption System Note 4: Direct Analysis of Spices and Coffee Note 3: Indoor Air Pollution Note 2: Detection of Arson Accelerants Using Dynamic Headspace with Tenax® Cartridges Thermal Desorption and Cryofocusing Note 1: Determination of Off-Odors and Other Volatile Organics In Food Packaging Films By Direct Thermal Analysis-GC-MS Tech No. "A" Note 14: Elimination of "Memory" Peaks in Thermal Desorption Improving Sensitivity in the H.P. 5971 MSD and Other Mass Spectrometers - Part I of II Improving Sensitivity in the H.P. 5971 MSD and Other Mass Spectrometers- Part II of II Adsorbent Resins Guide Development and Field Tests of an Automated Pyrolysis Insert for Gas Chromatography. Hydrocarbon Production in Pine by Direct Thermal Extraction A New Micro Cryo-Trap for the Trapping of Volatiles at the Front of a GC Capillary (019P) - Comparison of Septa by Direct Thermal Extraction Volatile Organic Composition in Blueberry Identification of Volatile Organic Compounds in Office Products Detection and Indentification of Volatiles in Oil Base Paintsby Headspace GC with On Column Cryo-Trapping Evaluation of Septa Using a Direct Thermal Extraction Technique INFLUENCE OF STORAGE ON BLUEBERRY VOLATILES Selection of Thermal Desorption and Cryo-Trap Parameters in the Analysis of Teas Redesign and Performance of a Diffusion Based Solvent Removal Interface for LC/MS The Design of a New Direct Probe Inlet for a Mass Spectrometer Analytes Using Mass Spectrometry (CRIMS) Application of SIMION 6.0 to Filament Design for Mass Spectrometer Ionization Sources A Student Guide for SIMION Modeling Software Application of SIMION 6.0 to Problems in Time-of-flight Mass Spectrometry Comparison of Sensitivity of Headspace GC, Purge and TrapThermal Desorption and Direct Thermal Extraction Techniques forVolatile Organics The Influence of Pump Oil Purity on Roughing Pumps Analysis of Motor Oils Using Thermal Desorption-Gas Chromatography-Mass Spectrometry IDENTIFICATION OF VOLATILE ORGANIC COMPOUNDS IN PAPER PRODUCTS Computer Modeling of Ion Optics in Time-of-Flight mass Spectrometry using SIMION 3D Seasonal Variation in Flower Volatiles Development of and Automated Microprocessor Controlled Gas chromatograph Fraction Collector / Olfactometer Delayed Extraction and Laser Desorption: Time-lag Focusing and Beyond A New Micro Cryo-Trap for the Trapping of Volatiles at the Front of a GC Column Design of a Microprocessor Controlled Short Path Thermal Desorption Autosampler Computer Modeling of Ion Optics in Time-of-Flight Mass Spectrometry Using SIMION 3D Thermal Desorption Instrumentation for Characterization of Odors and Flavors

- ▶
- 68. Using a Plug In UV-Vis Spectrometer to Monitor the Plasma Conditions in a GC CRIMS (EAS 98) (This Page)
1Scientific Instrument Services, Ringoes, NJ
2 Ocean Optics Inc., Dunddin,
FL 34698
BobK{at}OceanOptics.com
1998
INTRODUCTION
A low cost PC plug-in UV-Vis spectrometer is used to monitor the status of a microwave plasma for Chemical Reaction Interface Mass Spectrometry (CRIMS) [1]. The CRIMS cavity is placed within a gas chromatograph and connected to the spectrometer via a fiber optic. Examples are shown that illustrate the advantages of optical monitoring of the plasma and versatility of the plug-in system.
Background
The CRIMS method was developed at George Washington University in the early 1980s [1-3]. The technique replaces the need for radioactive tracers by measuring the ratios of stable isotopes. In order to distinguish isotopes of different mass from molecular species such as (M+H)+ etc., the analytes are broken down and mixed with a reactant gas in a microwave cavity. This destroys chemical information about the original molecule but generates the same simple products for all precursors. For example, analytes containing enriched 13C may be detected by reaction with SO2 to produce CO2. The ratio of 12CO2 (44 AMU) and 13CO2 (45 AMU) may then be monitored to selective identify 13C enriched analytes.
CRIMS Chemistry
Abramson [3] and others [4-6] have developed a variety of CRIMS chemistries for distinguishing between the isotopes of specific elements. These elements include: H, C, N, O, P, S, Cl, Se, and Br. The reactant gases employed include NF3, SO2, H2, N2, and HCl of which NF3 and SO2 appear to be the most versatile. Key to the selection of reactant gas is the generation of volatile and stable products whose masses do not overlap with other atomic or molecular species. It is preferable to generate products such as HF and DF which have unique masses in comparison to products such as 1H2H which requires high mass resolution to be distinguished from 1H3+.
Signals resulting from the natural abundance of the tracer isotope can easily be subtracted from the total signal to determine that part resulting solely from enrichment. For example, enriched 13CO2 signal can be found from 13CO2 = m/z 45 - 0.0119 * m/z 44. The factor 0.0119 accounts for both the natural 13C and 17O contributions.
CRIMS Advantages
CRIMS is intended to eliminate the need for radioactive tracers by determining the ratios of stable isotopes. Radioactive tracers are currently used in a variety of industries and have the advantage that other than a small change in mass the tagged species is not chemically modified. Their primary disadvantages are the need to produce and handle radioactive material and the dangers of introducing radioactive isotopes into living organisms. This is a severe limitation in Pharmacology and Biochemistry.
While radiochemical detection methods are normally considered highly sensitive, this is only true for off-line measurements where analysis time is not a limiting factor. In on-line measurements, as those made when analyzing chromatographic effluent, the detection time is limited to the width of the chromatographic peak and the detection limits are dramatically reduced. As a consequence, GC/CRIMS has been shown to produce substantially better signal to noise and/or resolution then on-line radiometric detection (for 14C) [3].

Figure 1. GC-CRIMS Cavity Mounted Inside a Hewlett-Packard Gas Chromatograph
Design of a GC-CRIMS System
A new CRIMS cavity is shown in Figure 1. This device is shown inside a GC oven for GC/CRIMS it could also be used after a desolvation apparatus for HPLC/CRIMS. In either case, a transfer line (fused silica capillary) is used to transport the reaction products to a mass spectrometer. Microwave power is supplied through a gold plated arm. The cavity is tuned via two finely threaded pole pieces located on opposite ends of the cavity. The (1/16" i.d.) reaction tube confines the chromatographic effluent and maintains a pressure sufficiently low to sustain a plasma. The plasma occurs only inside of this tube since the remainder of the cavity is at atmospheric pressure.
The CRIMS plasma is turned on immediately after the solvent slug has passed. Problems may occur at this point if: 1) the plasma fails to ignite or 2) the mixture of gasses within the reaction chamber is incorrect. In the past, the plasma status has been monitored by opening the GC oven and viewing the plasma directly with the eye. The plasma color was indicative of the gas mixture. This has the disadvantages that the GC oven temperature is perturbed and the color evaluation is subjective.
UV-Vis Spectrometer
A solution to the above problems is found through the use of a low cost optical spectrometer. Scientific Instrument Services, Inc. has incorporated a PC100 miniature fiber optic spectrometer from Ocean Optics, Inc. into the CRIMS system. The PC1000 is a low-cost 1024-element linear CCD-array fiber optic spectrometer (200-1000 nm wavelength range) mounted on a half-length, 500 kHz ISA-bus analog-to-digital converter card. The PC1000 conveniently installs into an ISA-bus slot in a PC and couples to fiber optics via a SMA 905 connector. In addition, multiple spectrometer channels can be easily connected to a PC1000 to expand wavelength range, perform multiple tasks or make reference measurements. The PC1000 comes with OOIBaseÔ operating software for WindowsÒ and SpectraScopeÔ operating software for DOS.

Figure 2. The PC1000 UV-Vis spectrometer From Ocean Optics
A critical element in the selection of a spectrometer from Ocean Optics was the OEM support provided. OEM's can create virtually any spectrometer configuration using the OEM board, one of three standard interface platforms (desktop, PC Plug-in and portable), 14 different grating choices, light sources and spectrophotometric accessories. This flexibility allows the OEM to optimize resolution, wavelength range and sensitivity within the desired spectral range. To provide optimal flexibility, Ocean Optics also offers customized spectroscopic components. For software integration, a series of *.dll files are available. These allow the integration of spectrometer software into a developers control system.
In the CRIMS system, light is collected after passing through the 3/32 inch wall of the ceramic reaction tube and directed to the spectrometer using a 2 meter fiber optic. The control software includes a user-friendly display for real-time observation and manipulation of the data. In the future, we expect the CRIMS control software to automatically detect the report the status of the plasma using the spectral data. The OEM support has made the incorporation of the spectrometer into CRIMS control software very simple but powerful.
Results
Figure 3 shows the data obtained using the OOIBase software. Figure 3A shows the OOIBase screen and a spectrum observed with just He carrier gas flowing through the cavity. Figures 3B and 3C, respectively, show the spectra recorded with too much SO2 reactant gas and with an organic contaminant. Figure 3D illustrates the data received under optimal plasma conditions. The intensities of the peaks observed are clearly indicative of plasma conditions. We can also detect the presence of excess gases, sample overloading, or air leaks. Use of the spectrometer has greatly simplified plasma observations and enhanced our ability to explain the interpretation of plasma conditions to users. In the future, output of the spectrometer software will communicate directly with the CRIMS electronics in order to automatically check plasma status and repeat ignition attempts when needed.
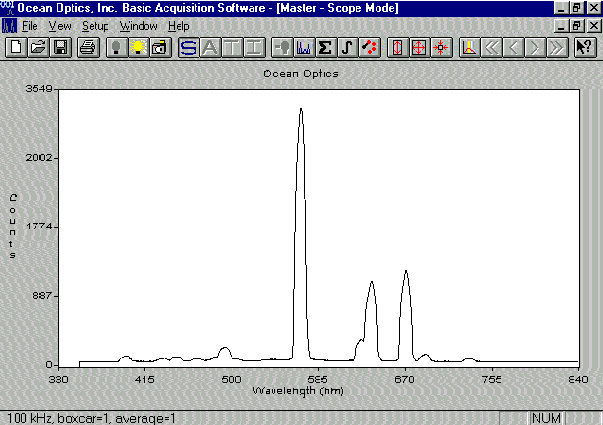
Figure 3a
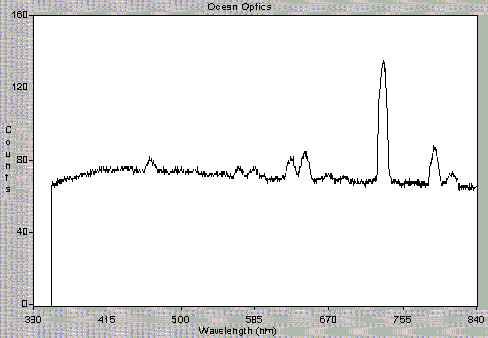
Figure 3b
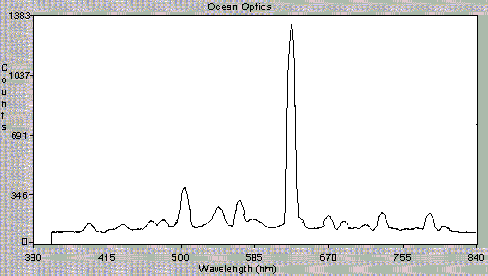
Figure 3c
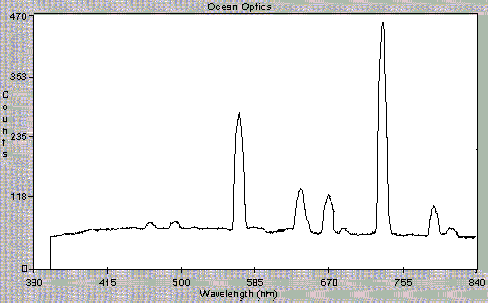
Figure 3d
Figure 3. Spectra observed under different CRIMS plasma conditions. Light is detected after passing through a 3/32 ceramic wall. A) He carrier gas, B) Too much SO2, C) Organic contamination, and D) Proper plasma conditions.
Conclusions
The PC1000 has proven to be a valuable addition to the CRIMS system. The support provided for OEM integration made the incorporation of the Ocean Optics product relatively simple. These factors have contributed to the development of significant improvements in the CRIMS product.
References
1. Abramson F. P.; Markey, S. P. Proc ASMS Conf. 1982, 30, 866-867.
2. Markey S. P.; Abramson F. P. Anal. Chem. 1982, 54, 2375-6.
3. Abramson F. P. Mass Spectrom. Rev. 1994, 13, 341-356.
4. Heppner, R. A. Anal. Chem.1983, 2170-2174.
5. Morre¢ , J. T.; Moini, M. Biol. Mass Spectrom. 1992, 21, 693-9.
6. Li, G.; Moini, M.; Rerez, F.; Ibarra, F. E.; Sandoval, D. Proc. ASMS Conf. 1994, 42, 293-4.

