- ▶
- Heaters/Source
- ▶
- Agilent Heaters and SensorsMass Spectrometry, Scientific Supplies & ManufacturingScientific Instrument Services 5973 Source Heater Tamper Resistant Allen Wrench 5973/5975 Quad Sensor 5985 Source Heater Assembly Agilent Interface Heater Assembly 5971 Interface Heater

- ▶
- Probes
- LiteratureApplication Notes Adsorbent Resins Guide Mass Spec Tips SDS Sheets FAQ MS Calibration Compound Spectra Manuals MS Links/Labs/ Organizations MS Online Tools Flyers on Products/Services Scientific Supplies Catalog About Us NextAdvance Bullet Blender® Homogenizer Protocols Micro-Mesh® Literature Instrumentation Literature Agilent GC/MS Literature SIS News / E-Mail Newsletter NIST MS Database - Update Notifications

- ▶
- Application NotesNote 103: EPA Method 325B, Novel Thermal Desorption Instrument Modification to Improve Sensitivity Note 102: Identification of Contaminants in Powdered Beverages by Direct Extraction Thermal Desorption GC/MS Note 101: Identification of Contaminants in Powdered Foods by Direct Extraction Thermal Desorption GC/MS Note 100: Volatile and Semi-Volatile Profile Comparison of Whole Versus Cracked Versus Dry Homogenized Barley Grains by Direct Thermal Extraction Note 99: Volatile and Semi-Volatile Profile Comparison of Whole vs. Dry Homogenized Wheat, Rye and Barley Grains by Direct Thermal Extraction GC/MS Note 98: Flavor and Aroma Profiles of Truffle Oils by Thermal Desorption GC/MS Note 97: Flavor Profiles of Imported and Domestic Beers by Purge & Trap Thermal Desorption GC/MS Note 96: Reducing Warping in Mass Spectrometer Filaments, with SISAlloy® Yttria/Rhenium Filaments Note 95: Detection of Explosives on Clothing Material by Direct and AirSampling Thermal Desorption GC/MS Note 94: Detection of Nepetalactone in the Nepeta Cataria Plant by Thermal Desorption GC/MS Note 93: Detection of Benzene in Carbonated Beverages with Purge & Trap Thermal Desorption GC/MS Note 92: Yttria Coated Mass Spectrometer Filaments Note 91: AutoProbe DEP Probe Tip Temperatures Note 90: An Automated MS Direct Probe for use in an Open Access Environment Note 89: Quantitation of Organics via a Mass Spectrometer Automated Direct Probe Note 88: Analysis of Silicone Contaminants on Electronic Components by Thermal Desorption GC-MS Note 87: Design and Development of an Automated Direct Probe for a Mass Spectrometer Note 86: Simulation of a Unique Cylindrical Quadrupole Mass Analyzer Using SIMION 7.0. Note 85: Replacing an Electron Multiplier in the Agilent (HP) 5973 MSD Note 84: Vacuum Pump Exhaust Filters - Charcoal Exhaust Traps Note 83: Vacuum Pump Exhaust Filters - Oil Mist Eliminators Note 82: Vacuum Pump Exhaust Filters Note 81: Rapid Bacterial Chemotaxonomy By DirectProbe/MSD Note 80: Design, Development and Testing of a Microprocessor ControlledAutomated Short Path Thermal Desorption Apparatus Note 79: Volatile Organic Compounds From Electron Beam Cured and Partially Electron Beam Cured Packaging Using Automated Short Path Thermal Desorption Note 78: A New Solution to Eliminate MS Down-Time With No-Tool-Changing of Analytical GC Columns Note 77: The Determination of Volatile Organic Compounds in VacuumSystem Components Note 76: Determination of the Sensitivity of a CRIMS System Note 75: An Apparatus for Sampling Volatile Organics From LivePlant Material Using Short Path Thermal Desorption Note 74: Examination of Source Design in Electrospray-TOF Using SIMION 3D Note 73: The Analysis of Perfumes and their Effect on Indoor Air Pollution Note 72: 1998 Version of the NIST/EPA/NIH Mass Spectral Library, NIST98 Note 71: Flavor Profile Determination of Rice Samples Using Shor tPath Thermal Desorption GC Methods Note 70: Application of SIMION 6.0 To a Study of the Finkelstein Ion Source: Part II Note 69: Application of SIMION 6.0 To a Study of the Finkelstein Ion Source: Part 1 Note 68: Use of a PC Plug-In UV-Vis Spectrometer To Monitor the Plasma Conditions In GC-CRIMS Note 67: Using Chemical Reaction Interface Mass Spectrometry (CRIMS) To Monitor Bacterial Transport In In Situ Bioremediation Note 66: Probe Tip Design For the Optimization of Direct Insertion Probe Performance Note 65: Determination of Ethylene by Adsorbent Trapping and Thermal Desorption - Gas Chromatography Note 64: Comparison of Various GC/MS Techniques For the Analysis of Black Pepper (Piper Nigrum) Note 63: Determination of Volatile and Semi-Volatile Organics in Printer Toners Using Thermal Desorption GC Techniques Note 62: Analysis of Polymer Samples Using a Direct Insertion Probe and EI Ionization Note 61: Analysis of Sugars Via a New DEP Probe Tip For Use With theDirect Probe On the HP5973 MSD Note 60: Programmable Temperature Ramping of Samples Analyzed ViaDirect Thermal Extraction GC/MS Note 59: Computer Modeling of a TOF Reflectron With Gridless Reflector Using SIMION 3D Note 58: Direct Probe Analysis and Identification of Multicomponent Pharmaceutical Samples via Electron Impact MS Note 57: Aroma Profiles of Lavandula species Note 56: Mass Spec Maintenance & Cleaning Utilizing Micro-Mesh® Abrasive Sheets Note 55: Seasonal Variation in Flower Volatiles Note 54: Identification of Volatile Organic Compounds in Office Products Note 53: SIMION 3D v6.0 Ion Optics Simulation Software Note 52: Computer Modeling of Ion Optics in Time-of-Flight mass Spectrometry Using SIMION 3D Note 51: Development and Characterization of a New Chemical Reaction Interface for the Detection of Nonradioisotopically Labeled Analytes Using Mass Spectrometry (CRIMS) Note 50: The Analysis of Multiple Component Drug Samples Using a Direct Probe Interfaced to the HP 5973 MSD Note 49: Analysis of Cocaine Utilizing a New Direct Insertion Probe on a Hewlett Packard 5973 MSD Note 48: Demonstration of Sensitivity Levels For the Detection of Caffeine Using a New Direct Probe and Inlet for the HP 5973 MSD Note 47: The Application Of SIMION 6.0 To Problems In Time-of-Flight Mass Spectrometry Note 46: Delayed Extraction and Laser Desorption: Time-lag Focusing and Beyond Note 45: Application of SIMION 6.0 to Filament Design for Mass Spectrometer Ionization Sources Note 44: The Design Of a New Direct Probe Inlet For a Mass Spectrometer Note 43: Volatile Organic Composition In Blueberries Note 42: The Influence of Pump Oil Purity on Roughing Pumps Note 41: Hydrocarbon Production in Pine by Direct Thermal Extraction Note 40: Comparison of Septa by Direct Thermal Extraction Note 39: Comparison of Sensitivity Of Headspace GC, Purge and Trap Thermal Desorption and Direct Thermal Extraction Techniques For Volatile Organics Note 38: A New Micro Cryo-Trap For Trapping Of Volatiles At the Front Of a GC Capillary Column Note 37: Volatile Organic Emissions from Automobile Tires Note 36: Identification Of Volatile Organic Compounds In a New Automobile Note 35: Volatile Organics Composition of Cranberries Note 34: Selection Of Thermal Desorption and Cryo-Trap Parameters In the Analysis Of Teas Note 33: Changes in Volatile Organic Composition in Milk Over Time Note 32: Selection and Use of Adsorbent Resins for Purge and Trap Thermal Desorption Applications Note 31: Volatile Organic Composition in Several Cultivars of Peaches Note 30: Comparison Of Cooking Oils By Direct Thermal Extraction and Purge and Trap GC/MS Note 29: Analysis Of Volatile Organics In Oil Base Paints By Automated Headspace Sampling and GC Cryo-Focusing Note 28: Analysis Of Volatile Organics In Latex Paints By Automated Headspace Sampling and GC Cryo-Focusing Note 27: Analysis of Volatile Organics In Soils By Automated Headspace GC Note 26: Volatile Organics Present in Recycled Air Aboard a Commercial Airliner Note 25: Flavor and Aroma in Natural Bee Honey Note 24: Selection of GC Guard Columns For Use With the GC Cryo-Trap Note 23: Frangrance Qualities in Colognes Note 22: Comparison Of Volatile Compounds In Latex Paints Note 21: Detection and Identification Of Volatile and Semi-Volatile Organics In Synthetic Polymers Used In Food and Pharmaceutical Packaging Note 20: Using Direct Thermal Desorption to Assess the Potential Pool of Styrene and 4-Phenylcyclohexene In Latex-Backed Carpets Note 19: A New Programmable Cryo-Cooling/Heating Trap for the Cryo-Focusing of Volatiles and Semi-Volatiles at the Head of GC Capillary Columns Note 18: Determination of Volatile Organic Compounds In Mushrooms Note 17: Identification of Volatile Organics in Wines Over Time Note 16: Analysis of Indoor Air and Sources of Indoor Air Contamination by Thermal Desorption Note 14: Identification of Volatiles and Semi-Volatiles In Carbonated Colas Note 13: Identification and Quantification of Semi-Volatiles In Soil Using Direct Thermal Desorption Note 12: Identification of the Volatile and Semi-Volatile Organics In Chewing Gums By Direct Thermal Desorption Note 11: Flavor/Fragrance Profiles of Instant and Ground Coffees By Short Path Thermal Desorption Note 10: Quantification of Naphthalene In a Contaminated Pharmaceutical Product By Short Path Thermal Desorption Note 9: Methodologies For the Quantification Of Purge and Trap Thermal Desorption and Direct Thermal Desorption Analyses Note 8: Detection of Volatile Organic Compounds In Liquids Utilizing the Short Path Thermal Desorption System Note 7: Chemical Residue Analysis of Pharmaceuticals Using The Short Path Thermal Desorption System Note 6: Direct Thermal Analysis of Plastic Food Wraps Using the Short Path Thermal Desorption System Note 5: Direct Thermal Analysis Using the Short Path Thermal Desorption System Note 4: Direct Analysis of Spices and Coffee Note 3: Indoor Air Pollution Note 2: Detection of Arson Accelerants Using Dynamic Headspace with Tenax® Cartridges Thermal Desorption and Cryofocusing Note 1: Determination of Off-Odors and Other Volatile Organics In Food Packaging Films By Direct Thermal Analysis-GC-MS Tech No. "A" Note 14: Elimination of "Memory" Peaks in Thermal Desorption Improving Sensitivity in the H.P. 5971 MSD and Other Mass Spectrometers - Part I of II Improving Sensitivity in the H.P. 5971 MSD and Other Mass Spectrometers- Part II of II Adsorbent Resins Guide Development and Field Tests of an Automated Pyrolysis Insert for Gas Chromatography. Hydrocarbon Production in Pine by Direct Thermal Extraction A New Micro Cryo-Trap for the Trapping of Volatiles at the Front of a GC Capillary (019P) - Comparison of Septa by Direct Thermal Extraction Volatile Organic Composition in Blueberry Identification of Volatile Organic Compounds in Office Products Detection and Indentification of Volatiles in Oil Base Paintsby Headspace GC with On Column Cryo-Trapping Evaluation of Septa Using a Direct Thermal Extraction Technique INFLUENCE OF STORAGE ON BLUEBERRY VOLATILES Selection of Thermal Desorption and Cryo-Trap Parameters in the Analysis of Teas Redesign and Performance of a Diffusion Based Solvent Removal Interface for LC/MS The Design of a New Direct Probe Inlet for a Mass Spectrometer Analytes Using Mass Spectrometry (CRIMS) Application of SIMION 6.0 to Filament Design for Mass Spectrometer Ionization Sources A Student Guide for SIMION Modeling Software Application of SIMION 6.0 to Problems in Time-of-flight Mass Spectrometry Comparison of Sensitivity of Headspace GC, Purge and TrapThermal Desorption and Direct Thermal Extraction Techniques forVolatile Organics The Influence of Pump Oil Purity on Roughing Pumps Analysis of Motor Oils Using Thermal Desorption-Gas Chromatography-Mass Spectrometry IDENTIFICATION OF VOLATILE ORGANIC COMPOUNDS IN PAPER PRODUCTS Computer Modeling of Ion Optics in Time-of-Flight mass Spectrometry using SIMION 3D Seasonal Variation in Flower Volatiles Development of and Automated Microprocessor Controlled Gas chromatograph Fraction Collector / Olfactometer Delayed Extraction and Laser Desorption: Time-lag Focusing and Beyond A New Micro Cryo-Trap for the Trapping of Volatiles at the Front of a GC Column Design of a Microprocessor Controlled Short Path Thermal Desorption Autosampler Computer Modeling of Ion Optics in Time-of-Flight Mass Spectrometry Using SIMION 3D Thermal Desorption Instrumentation for Characterization of Odors and Flavors

- ▶
- Note 66: Probe Tip Design For the Optimization of Direct Insertion Probe Performance (This Page)
Presented at ASMS, Orlando, FL, CA., June 1998
INTRODUCTION
The direct probe has been used for many years to introduce samples into the mass spectrometer. It has the advantages of permitting a quick analysis with minimal sample preparation. However, it has the disadvantage of making the identification of mixtures of compounds difficult due to the complexity of the resulting mass spectrum when multiple compounds are analyzed. Many users have overcome this obstacle utilizing the chemical ionization mass spec technique. This produces simplified spectra which can be interpreted with multiple component samples. However, the simplified spectra do not provide a conclusive identification of the analytes.
The purpose of this study is to evaluate the design of the direct probe tip and the direct probe sample vial to improve the direct probe technique. Different style probe tips as well as probe tips made from different materials were studied. Both the sensitivity of the technique for different styles of probe tips as well as the ability of the technique to separate multiple components in a single sample were studied. In addition, it would be beneficial to develop a probe tip that can analyze samples that could not be analyzed by other techniques due to the thermal instability of the compounds, high molecular weight or their inability to be chromatographed.
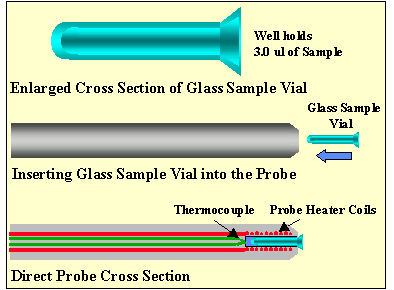
Figure 1 - Glass Sample Vial For the HP5973 Direct Insertion Probe
The standard probe uses flared glass sample vials (1.7mm diameter by 9 mm long) into which the sample is inserted. The sample vial is inserted into the probe tip where it is held in place by a small spring (Figure # 1). The glass sample vial is the standard vehicle for introduction of samples into the mass spectrometer. Solid matrix samples can be easily inserted into the glass sample vial for subsequent analysis. Most frequently, samples are dissolved or extracted into a volatile solvent, as methanol or chloroform and is then injected into the sample vial using a standard microliter syringe. The solvent is then evaporated to dryness, leaving the analyte residue in the glass vial for analysis. The glass sample vials can hold up to 3 uL of sample. The disadvantage of this technique is the design of the sample vial. The vial is quite deep, thereby making it difficult to volatilize the entire sample at the same time and introduce the volatile sample into the mass spec source in short time period. It is anticipated that the probe technique could be improved by making this vial cavity smaller and constructing the sample vial out of a material that would heat more quickly. These two design improvements would introduce the sample into the mass spec source in a shorter period of time, thereby improving the sensitivity of the technique.
Experimental
The Scientific Instrument Services Direct Insertion Probe was used in conjunction with the Hewlett Packard 5973 MSD. The mass spec probe tip can be heated ballistically up to 450oC at a ramp rate in excess of 400o per minute. The probe can also be temperature programmed for slower heating applications. The HP 5973 MSD was operated in the El mode and was scanned front mass 50 to 350 daltons. The mass spec was scanned at 1 scan per second. The GC control systems were disconnected via the software controls, and the analysis time was set for 3.0 minutes. After the sample analysis was complete, the total ion chromatogram was viewed using the ChemStation data analysis module. The resulting mass spectrum was recorded.
For this study, a mixture of the sugars fructose and sucrose were used. These two sugars were mixed and dissolved in methanol. This solution was then injected into the various probe tips for analysis. These sugars can not normally be analyzed via the direct probe technique using glass sample vials. The methanol solvent in each of the sample vials was allowed to evaporate to dryness before analyzing any of the samples. This was accomplished in about 30 seconds by allowing the solvent to evaporate at room temperature. Each sample was then inserted into the mass spec probe and inserted through the vacuum lock system of the probe inlet into the mass spec source of the HP 5973 MSD. Each sample was then heated ballistically, and the mass spec was scanned to analyze the thermally extracted sample. The total analysis time was 2 minutes or less per sample. As an alternative technique, the direct probe was temperature ramp at rates of 50 to 500o per minute.
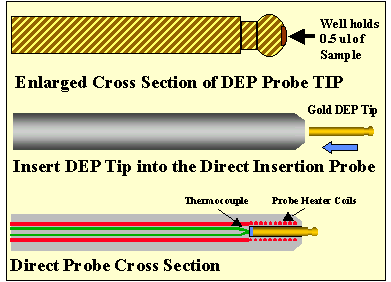
Figure 2 - Gold Plated DEP Probe TIP For the HP5973 Direct Insertion Probe
The fist probe tip was constructed out of a copper rod. The copper rod fit snugly into the probe tip and was made long enough to extend fully into the direct probe cavity and extent out about 2.0 millimeters. The copper probe tip was tapered at the end protruding out of the probe, and a small well (0.03" diameter x 0.30" deep) was drilled into the tip (Figure # 2). Copper was chosen due to its ability to heat quickly. The small sample well holds less than 0.5 uL of sample. This probe tip was then tested by injecting the sugar sample into the sample well and then heating it ballistically to various temperatures from 250o C to 450o C. Next, the probe tip was studied by heating it at different ramp rates from 50o per minute to 500o per minute.
A final probe tip was constructed from a solid gold rod. It was shaped the same as the copper tip above (.Figure # 2). Studies were conducted using this probe tip, as were done for the copper probe tip above. Additional studies were done on the length of the probe tip to see how this effected the sensitivity of the technique.
Results and Discussion
The copper tip proved to be quite satisfactory for the analysis of the sugar samples (Figure #3). The analysis of the sugar mixture produced two distinct peaks with distinctive mass spectra for each sugar. Similar results were obtained using mixtures of pharmaceuticals in previous studies of the HP 5973 probe using glass sample vials. However, this technique proved to be much faster and more sensitive than achieved using glass sample vials. The sample was contained in a smaller well with less surface area that was heated more quickly. The resulting total ion chromatograph produced peaks that had less peak width and were much higher, therefore, increased sensitivity.
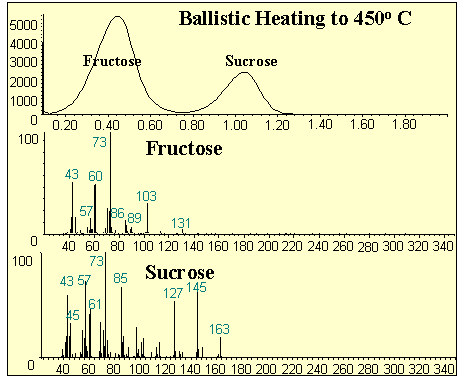
Figure 3 - Analysis of Sugars Via MSD-DEP Probe Tip
In the first series of tests, the probe tip was ballistically heated to different temperatures from 250o C to 450o C (Figure # 4). The higher the temperature, the faster the analytes were desorbed off the probe tip producing less band width and greater sensitivity. Only a minor loss in resolution occurred at the higher temperatures. No decomposition of the sample was apparent at the higher temperatures.
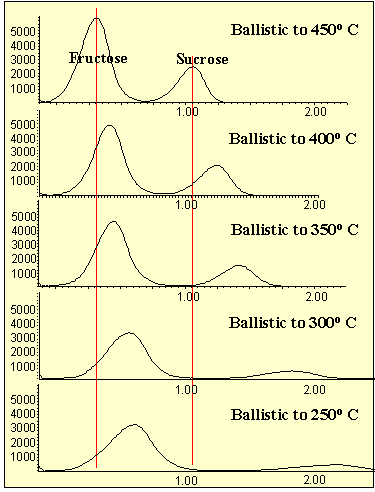
Figure 4 - Analysis of Sugars Via Copper MSD-DEP Probe Tip
The second series of tests studied the copper tip to determine the effect of ramp rate. The new HP5973 probe was designed to ramp at high rates (greater than 500o per minute). In addition, it can be temperature programmed when this is required. The sugar samples were ramped at various rates from 50 to 500o per minute ( Figure # 5 ). These were compared to the ballistic heating of the probe tip described above. Higher ramp rates produced less band width and greater sensitivity. The ballistic method was still the most sensitive method of analysis.
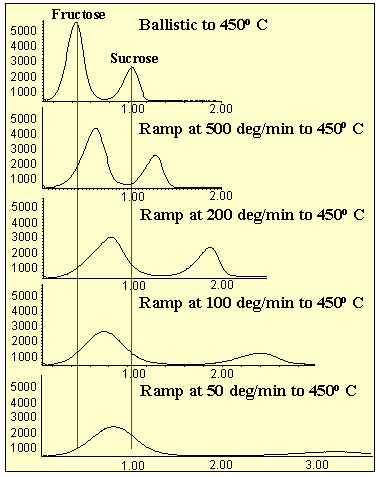
Figure 5 - Analysis of Sugars Via Copper MSD-DEP Probe Tip
The gold probe tip was studied using the same sugar sample and studied as above. The gold tip produced even better results. Heating the gold tip ballistically to 450o C produced even less band width and greater sensitivity than the copper probe tip (Figure # 6). This is probably due to the better heat conduction of gold over copper. Additional studies were conducted on the length of the gold probe tip that extends from the probe rod. Probe tips that extend further from the probe rod and into the mass spec source lost some of the heating capacity the probe rod. A probe tip that extended 1.0 to 2.0 millimeters from the tip of the probe provided optimal results.
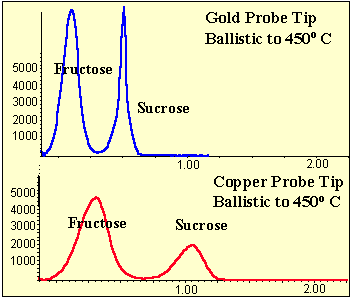
Figure 6 - Comparison of Gold and Copper DEP Probe Tip
Conclusion
The heated direct probe sample introduction system has proven to be a useful addition to new Hewlett Packard 5973 MSD for the analysis of solid matrix samples. Samples were analyzed in less than 1.5 minutes using the ballistic heating probe method. However, the probe can be temperature programmed at slower rates for the purpose of thermally separating mixtures of drugs in a single sample. The direct probe analysis of samples has proven to be a sensitive technique for solid sample analysis with detection levels down to 10 ng. The peak width is quite large as compared to a GC analysis and explains the loss in sensitivity compared to conventional GC analysis, where peak widths are normally less than 5 seconds. The highly sensitive HP 5973 MSD and the versatile ChemStation software permit the analysis of multiple component samples via the El mass spec probe technique to achieve fast and definitive method of analysis. The direct probe introduction system interfaced to HP 5973 will be a valuable technique for the analysis of many drug and pharmaceutical samples, where fast sample analysis is required.
The direct probe technique for the HP 5973 MSD has been improved throuh the design new gold sample vial holder. The gold probe tip used in the ballistic mode produced the best results of the various tips studied. This new tip had a smaller well for the sample with less surface area, which could be heated more rapidly - thereby producing greater sensitivity. Using these new gold probe tips, the sensitivity of the direct probe was increased 2 to 20 times over direct probe technique using glass sample vials. In addition, samples as sugars, high boiling pesticides and pharmaceuticals can be analyzed. Previously, these samples were difficult or impossible to analyze via the direct probe. In addition, mixtures of analytes in the same sample can also be analyzed. This analysis of mixtures is enhanced by the use of the HP ChemStation software to separate and identify the multiple component samples.
Additional work still needs to be done with this technique such as analyzing additional compounds and mixtures of compounds. The reproducibility of the technique also needs to be studied and improved. In addition, glass sample vials with smaller sample wells need to be studied and compared to the gold probe tips. The disadvantage of the gold tips is their cost. The tips must be cleaned and reused, but the glass sample vials are inexpensive and disposable.


